Chemistry and Chemists № 1 2026
Journal of Chemists-Enthusiasts
| Content | Chemistry experiments - video | Physics experiments - video | Home Page - Chemistry and Chemists |
|
Chemistry and Chemists № 1 2026 Journal of Chemists-Enthusiasts |
Freezing of Supercooled Water - pt.3, 4 Chemist |
|
Having noticed a mistake in the text, allocate it and press Ctrl-Enter
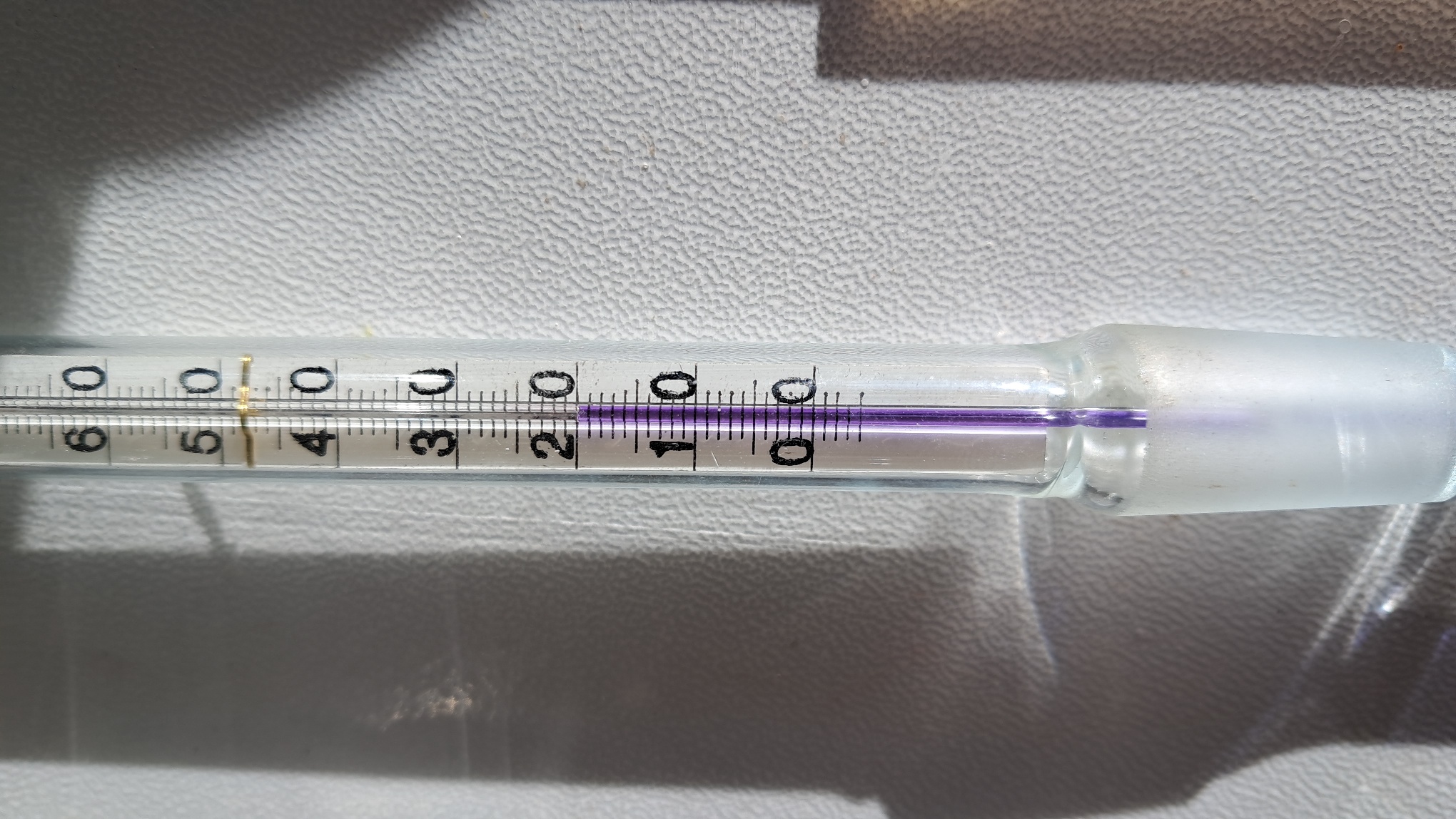
Let us continue our inventory of frozen liquids in the cold laboratory. Buffer solutions for calibrating a pH meter stood on a shelf, alongside a sodium chloride solution and a mixed solution of sodium carbonate and sodium chloride. The buffer solution with a pH of 9.18 froze first, and a few days later the pH 4.01 solution solidified as well. Since these solutions were stored in plastic bottles, the freezing caused no damage. Plastic containers usually withstand the freezing of aqueous solutions without breaking. The dilute sodium chloride solution (6%) and the solution containing sodium carbonate (3%) with sodium chloride (6%) remained liquid throughout the winter.
The saturated solutions of potassium dihydrogen phosphate and potassium nitrate had still not frozen. The dihydrogen phosphate crystals were visually striking, whereas the nitrate crystals were rather unremarkable. One night at home, I suddenly remembered that three 250 mL glass volumetric flasks containing pH buffer solutions were still in the laboratory. If those solutions froze, the flasks would crack. Unlike plastic bottles, glass vessels filled with aqueous solutions often shatter when the contents freeze. I went to the laboratory first thing the next morning. Two of the buffer solutions remained liquid, but the pH 9.18 buffer had frozen and the flask had cracked. Fortunately, the frozen solution retained its shape, so I carefully placed it in a beaker and brought it to the heated laboratory to thaw. That room was exposed to direct sunlight, and despite the bitter cold outside, the temperature inside rose to 20°C. Some houseplants had even begun to bloom. The ice gradually melted, and I transferred the buffer solution into another flask. A little later, my colleague arrived. Seeing the frozen buffer solution and the cracked flask, he suddenly remembered that several desiccators filled with tritiated water were stored in his laboratory. In addition to the laboratory he had turned into a storage room, he had another room - an unheated laboratory designed for working with tritium. Tritium experiments were conducted there when the room was warm, of course, but now the temperature had also dropped below freezing. The question was: what had happened to the desiccators filled with tritiated water? |

Frozen pH Buffer Solutions |

|

|

|

|

|

|

|

|

|

|

|

|
|

|

|

|

|

|

|

|

Chemical laboratory for tritium research (in the warm season) |

|

|

|

These algae accidentally grew in tritiated water |

|

|

|

|

|

|

|

|

|

|

|
|

|

''Frozen'' Chemical Laboratory |

|

|

|

|

|

|

|

|

|

Freezing of Supercooled Water |
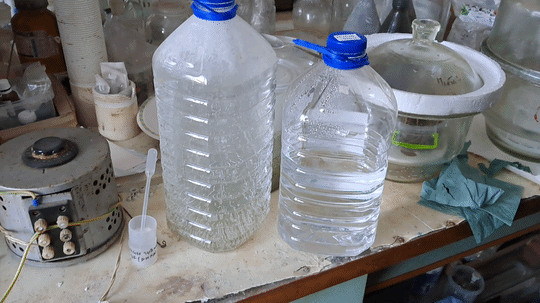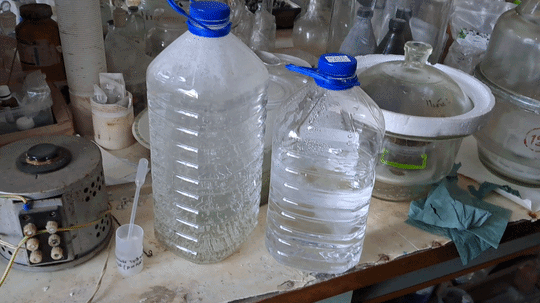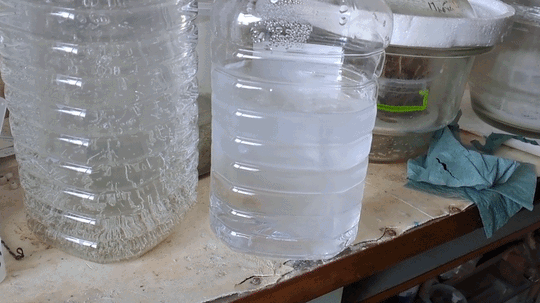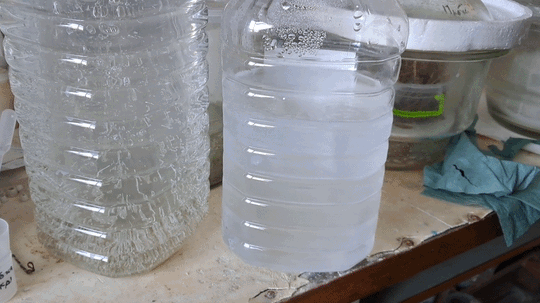
|