Chemistry and Chemists № 1 2026
Journal of Chemists-Enthusiasts
| Content | Chemistry experiments - video | Physics experiments - video | Home Page - Chemistry and Chemists |
|
Chemistry and Chemists № 1 2026 Journal of Chemists-Enthusiasts |
Freezing of Supercooled Water - pt.5, 6 Chemist |
|
Having noticed a mistake in the text, allocate it and press Ctrl-Enter
I went to the unheated laboratory and immediately checked the thermometer. The temperature was -5°C. I secured my smartphone to a tripod and started recording.
I carefully shook the first bottle of distilled water; however, the water did not begin to freeze. Then I picked up the second bottle - the one that had already frozen - and discovered that some of the water inside it was still liquid. In other words, it had not completely frozen while standing in the cold room. I shook the third and fourth bottles of distilled water - the supercooled water in them remained liquid. I opened the cap of the first bottle and shook it again. This time, the liquid began to crystallize rapidly. Suddenly, I noticed that the water in the fourth bottle had also started to freeze. About a minute later, white, opaque ice formed inside both the first and fourth bottles. However, the water in the third bottle was still liquid. I shook it vigorously. Rapid crystallization began, and soon all the water turned into white ice. There was still one full bottle and one partially filled bottle of supercooled water left. I turned off the camera, moved the smartphone closer, carefully adjusted the focus, and shook the full bottle. The water froze quickly - the experiment was perfect. I was delighted - and only then realized that I had forgotten to turn on the video recording. One six-liter bottle remained, containing only about one liter of distilled water. I shook it, and the water froze within seconds. So, I did manage to record the instant freezing of supercooled water. Unfortunately, there was a fly in the ointment: when I attempted to film the process close up, the camera had been turned off. I was deeply disappointed. The question was: could the experiment be repeated? At the time, I did not know the answer, but I decided to try. I brought a fresh bottle of distilled water into the cold laboratory and left it there overnight. I hoped the water would supercool and remain liquid, as had happened with the previous five bottles. However, those bottles had cooled gradually along with the room. Now the situation was different - I had brought the bottle into an already cold laboratory, so I was unsure whether the water would remain liquid or freeze by morning. To increase my chances, I also took one of the frozen bottles into the warm laboratory. It would thaw overnight and could then be returned to the cold room for another attempt. The next day, I found that the water in the bottle left in the cold laboratory had indeed remained liquid. I turned on the camera, shook the bottle, and the contents froze. I noticed that although the freezing began almost instantly, it was not complete: some of the water remained liquid. The bottle did not contain solid ice, but rather a slushy mixture. Had I understood the significance of this observation at the time, I would have spared myself considerable frustration later - but I will not get ahead of the story. For comparison, I shook the bottle of distilled water that I had just brought back from the warm laboratory. As expected, the water remained liquid. |

Freezing of Supercooled Water |

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|
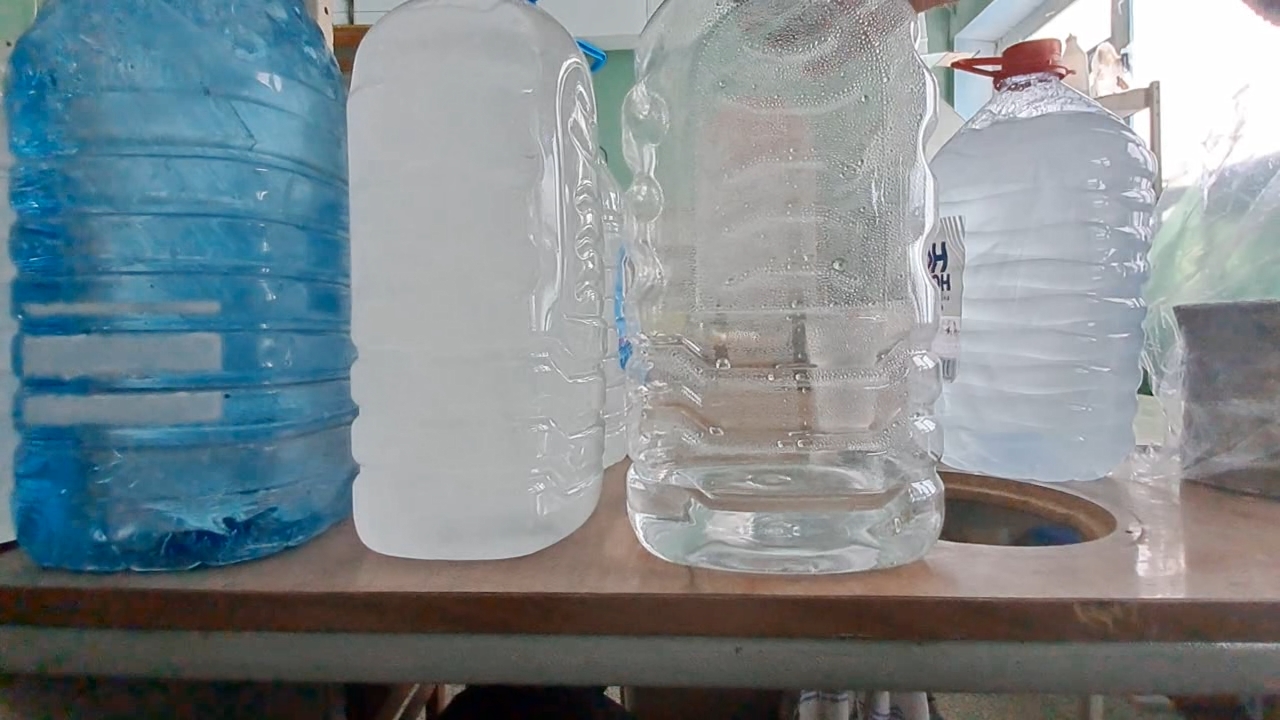
|
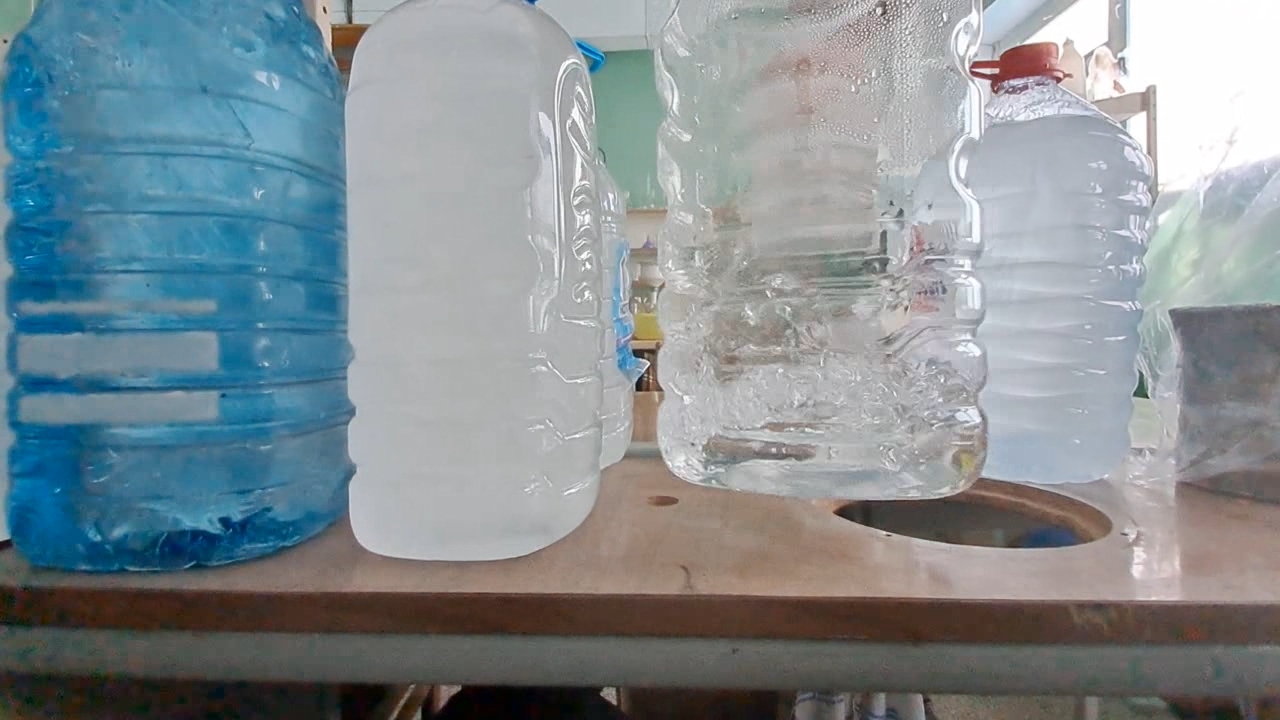
|

|

|

|

|

|

|

|

|

|

Speed x2 |

|

|

Water Volume Shrinkage When Heating |

|

|

|

|
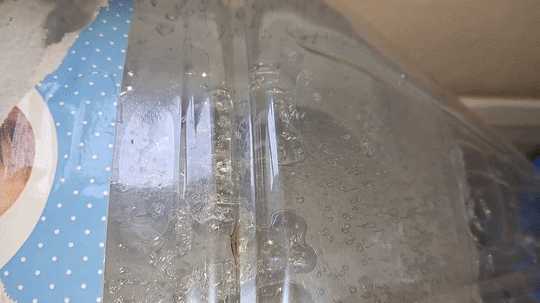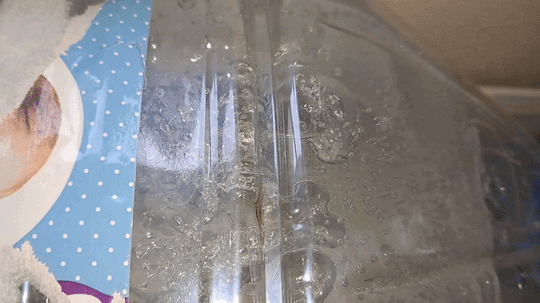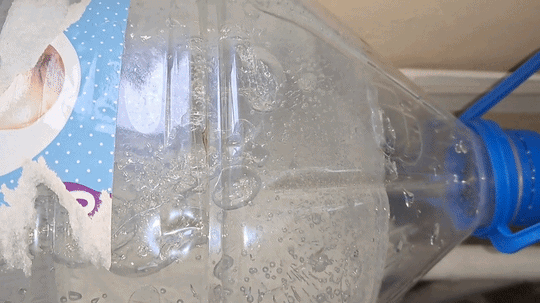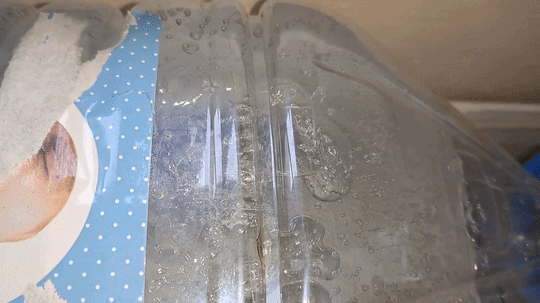
|

|

|