Chemistry and Chemists № 1 2026
Journal of Chemists-Enthusiasts
| Content | Chemistry experiments - video | Physics experiments - video | Home Page - Chemistry and Chemists |
|
Chemistry and Chemists № 1 2026 Journal of Chemists-Enthusiasts |
Experiments with Universal Indicator - pt.6, 7 Chemist |
|
Having noticed a mistake in the text, allocate it and press Ctrl-Enter
Color Change of Universal pH Indicator - Part 6
For greater clarity, I conducted the experiment in a tall 1-liter beaker. I poured approximately 700 mL of tap water into the beaker, turned on the magnetic stirrer, and increased the speed. An air vortex formed in the beaker, extending down to the bottom (or rather, to the magnetic stir bar).
Изменение окраски универсального индикатора pH - Часть 6 I added a few drops of the brown alcoholic solution of the universal indicator directly into the air "funnel" of the vortex. At first, the water surrounding the funnel turned brown. However, as a result of stirring, the entire solution soon became green. I then began adding 13% hydrochloric acid dropwise. The solution gradually changed color: from green to yellow, from yellow to orange, and then from orange to red-orange. To lower the pH as much as possible, I added 69% nitric acid; the liquid in the beaker turned crimson. After this, I began adding 25% ammonia solution dropwise. Near the vortex, the solution developed a localized blue coloration, which quickly disappeared upon stirring. The color of the bulk solution gradually changed in reverse order: crimson, red, orange, yellow, green, and blue. To further increase the pH, I added sodium hydroxide granules to the beaker. The solid dissolved slowly, and the indicator turned violet. I then lowered the pH again by adding 69% nitric acid in small portions. A gradual color change occurred in reverse order: violet, blue, green, yellow, orange, red, and crimson. The addition of acid and alkali can be alternated multiple times, causing the indicator to change color in opposite directions. If the acid or alkali is added slowly enough (or if more dilute solutions are used), not only distinct colors but also a range of intermediate shades can be observed. A veritable rainbow appears in the beaker, accompanied by a tornado-like vortex. |

Color Change of Universal pH Indicator |

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|
|
Having noticed a mistake in the text, allocate it and press Ctrl-Enter
Universal Indicator Paper, Hydrochloric Acid, and Ammonia - Part 7
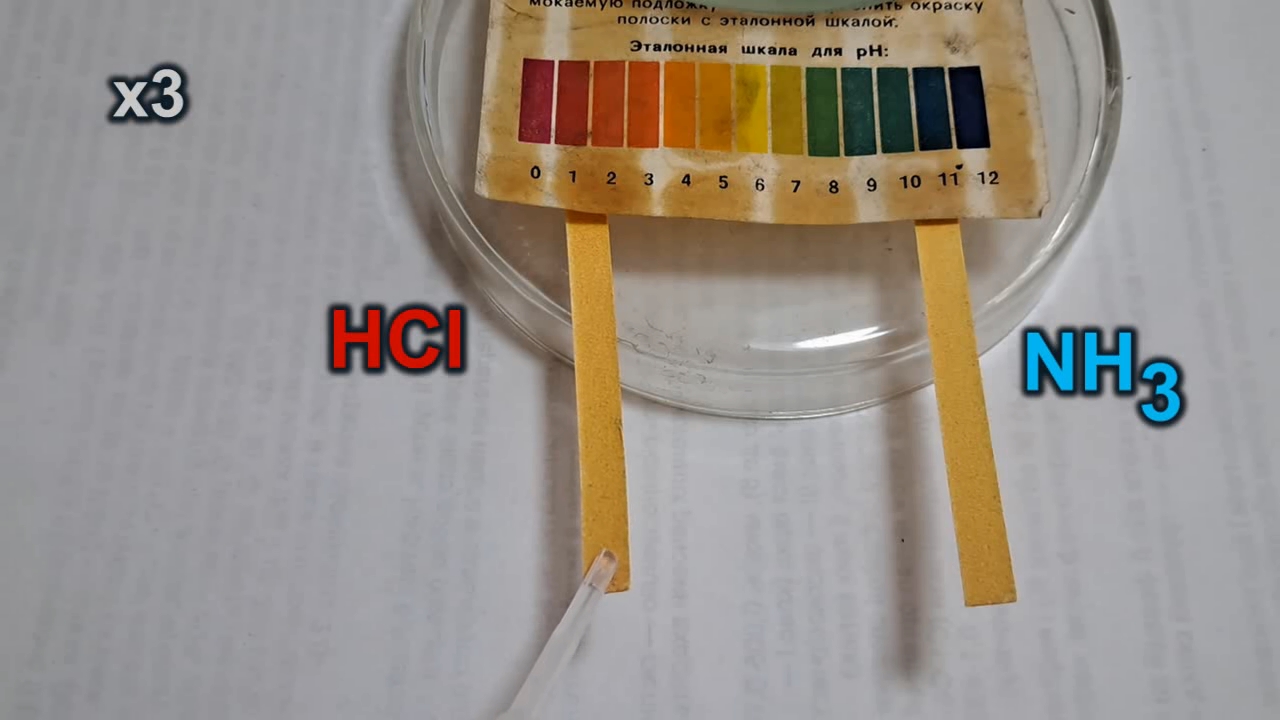
Chemists frequently encounter universal indicator paper. It is used to quickly estimate the pH of a solution, especially when a pH meter is not available. The procedure is very simple: the tip of a paper strip is dipped into the solution being tested and immediately removed. The resulting color is then compared with a reference chart. The accuracy of this method is limited; therefore, in most cases, universal indicator paper is used simply to determine whether a solution is acidic or alkaline.
Универсальная индикаторная бумага, соляная кислота и аммиак - Часть 7 After use, a strip of indicator paper is usually discarded. Some chemists tear off the wetted portion and reuse the remaining part. This practice is often explained by the need to conserve materials: many scientific institutions in our country do not allocate sufficient funding for reagents and equipment. Even when funds are allocated, they are often misused. At first glance, the properties of universal indicator paper seem well known: it turns red in an acidic medium and blue in an alkaline one. It might therefore appear unlikely that any interesting experiments could be devised using it. However, the situation is not so simple. For a long time, I have been considering the following question. Suppose one strip of universal indicator paper is wetted with a volatile acid and another with a volatile base. The first strip will turn red, and the second blue - so far, everything is predictable. Let's leave both strips and allow the solutions to evaporate gradually. What will happen? It seems logical to assume that the acid and base will evaporate, and both strips will gradually return to their original yellow color. It is also reasonable to expect that intermediate colors may appear during evaporation. The answer appears obvious. However, chemistry is an experimental science. Mere speculation - like the well-known "quantum parable" involving a radioactive atom, a box, and an unfortunate cat - is not sufficient. Even an apparently obvious assumption must be verified experimentally. 
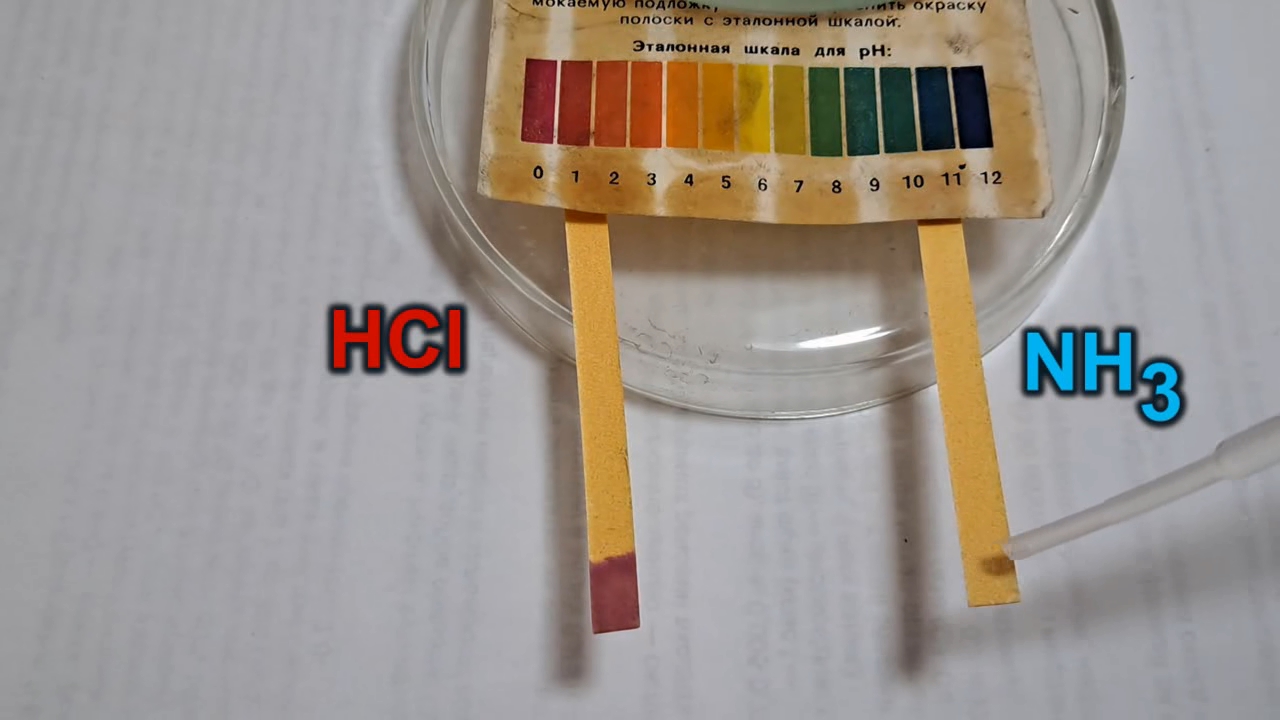
Schrodinger's cat is alive and very angry I touched the first strip with a pipette containing a 13% hydrochloric acid solution. The end of the strip turned crimson, and the colored zone slowly extended along the paper with the advancing liquid front. I then brought a pipette containing a 25% ammonia solution toward the second strip. Even before contact, the indicator began to turn green. Upon touching the paper, the indicator turned blue. However, the ammonia solution partially dissolved the organic dyes and washed them out of the paper, producing a pale spot within the blue region. This phenomenon was reminiscent of paper chromatography. In the case of the acid, no such pale spot was observed - the end of the strip was uniformly colored crimson, forming a clear boundary with the yellow portion that had not been exposed. On the strip wetted with ammonia, the blue color gradually faded. It then turned green, and eventually brown. The strip did not return to its original color because the ammonia solution had dissolved and removed part of the indicator from the paper. In contrast, on the first strip, the crimson region not only failed to fade but continued to expand slowly. Although the liquid front had stopped moving, the expansion was caused by the action of hydrochloric acid vapor. A diffuse crimson coloration appeared on the previously yellow portion of the strip. After this, I left the strips unattended for some time, as I did not expect further significant changes. However, after about 20 minutes, I found that the second strip had also partially turned crimson - the very strip that had previously been wetted with ammonia. The coloration was uneven and diffuse: hydrochloric acid vapor had affected not only the strip to which the acid had been applied but also the adjacent strip. This effect intensified over time. After 24 hours, the crimson color had noticeably faded but had not disappeared. I repeated the experiment and obtained similar results. Incidentally, this time I secured the indicator strips with adhesive tape, so there was no risk of displacing them accidentally with a pipette. This reminded me of physicist colleagues who were assembling a complex optical system requiring precise alignment; to fix the components in place, they used… children's plasticine. Let us summarize. Prediction. A strip of universal indicator paper wetted with hydrochloric acid will turn red. As the acid evaporates, the paper will return to its original color. Similarly, a strip wetted with ammonia will turn blue and, after evaporation, return to its original color. Observations. One strip turned crimson upon contact with hydrochloric acid. The other turned blue upon contact with ammonia; partial dissolution and leaching of the dye were observed. As the ammonia evaporated, the second strip changed color from blue to green and then to brown. The color of the first strip remained unchanged for a long time; moreover, due to hydrochloric acid vapor, both strips developed a diffuse crimson coloration in areas not exposed to the liquid acid. After 24 hours, the crimson color faded but did not disappear. As can be seen, the results of this simple experiment do not fully agree with the initial prediction. |

Universal Indicator Paper, Hydrochloric Acid, and Ammonia |
|
|

|

|

|

|

|

|

|

|

|

|

|

|

|

|