Chemistry and Chemists № 1 2026
Journal of Chemists-Enthusiasts
| Content | Chemistry experiments - video | Physics experiments - video | Home Page - Chemistry and Chemists |
|
Chemistry and Chemists № 1 2026 Journal of Chemists-Enthusiasts |
Experiments with Universal Indicator - pt.10, 11 Chemist |
|
Having noticed a mistake in the text, allocate it and press Ctrl-Enter
Universal Indicator, Phenolphthalein, and Sodium Hydroxide - Part 10
I slightly modified the final part of the previous experiment in order to obtain the desired result when treating phenolphthalein paper with a saturated sodium hydroxide solution.
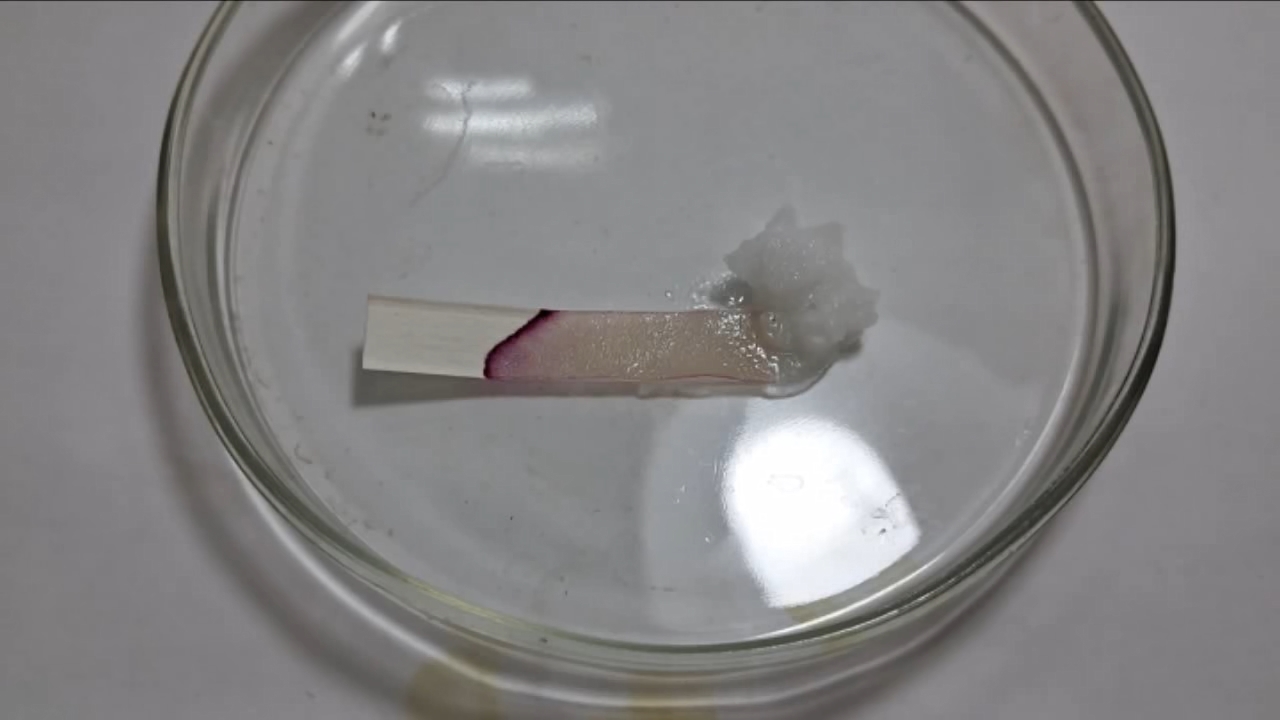
Универсальный индикатор, фенолфталеин и гидроксид натрия - Часть 10 I placed several sodium hydroxide granules in a Petri dish and moistened them with water. After waiting about a minute for the alkali to dissolve partially, I touched the end of a strip of phenolphthalein paper to the sodium hydroxide. The end of the strip immediately turned a dark fuchsia color. The fuchsia stain began to spread across the paper. Once again, the saturated alkali solution had colored the paper fuchsia! I said to a colleague who was observing the indicator paper with me: "It didn't work again." My colleague left, and I continued observing. The dark fuchsia region spread across the paper. Soon, the part of the colored area that had been in direct contact with the alkali began to fade. The fuchsia color weakened, and the paper slowly turned white. Before long, only the advancing liquid front retained its bright fuchsia color. This time, I realized that the discoloration of the paper was not due to the leaching of the phenolphthalein but rather to the decolorization of the indicator. This meant that upon contact with a highly alkaline solution, phenolphthalein first turns fuchsia, forming the familiar Ind2- anion, which then slowly transforms into the colorless Ind(OH)3- anion. Recall that during the titration of an acid with an alkali, the colorless form of phenolphthalein, H2Ind, transforms into the fuchsia form, Ind2-. This transformation occurs essentially instantaneously, which is essential for successful titration. In contrast, the transformation of the fuchsia form Ind2- into colorless Ind(OH)3- in a highly alkaline solution took several minutes. The liquid front had practically stopped. I rotated the strip of phenolphthalein paper by 180 degrees so that the alkali contacted the opposite end, in order to achieve complete loss of color. Upon contact with the alkali, the paper again turned bright fuchsia, which gradually faded. After a few minutes, the strip became white. I then touched the alkali with a strip of universal indicator paper, and a small dark green spot appeared on the yellow paper. Most of the added water had already been absorbed by the strip of phenolphthalein paper, so the liquid front on the universal indicator strip quickly stopped moving. I added a little more water to the solid alkali, and the green-colored zone spread across the entire strip. While adding the water, an interesting thought occurred to me. Phenolphthalein is colorless in concentrated alkali solutions and fuchsia in dilute ones. Therefore, if white phenolphthalein paper soaked with saturated sodium hydroxide solution were placed in a small amount of water, the alkali concentration would decrease, and a fuchsia color should reappear. I added water to the Petri dish containing the strip of phenolphthalein paper and shook the contents, but no color appeared. I then carefully added a few drops of 70% acetic acid to partially neutralize the alkali and shook the Petri dish again. A faint pink color appeared on the phenolphthalein paper. I stopped the experiment, washed the Petri dish, and the next day found the strip of phenolphthalein paper in the sink. It had turned a distinct fuchsia. |

Universal Indicator, Phenolphthalein, and Sodium Hydroxide |

|

|

|

|

|

|

|

|

|
|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

The next day |