Chemistry and Chemists № 1 2026
Journal of Chemists-Enthusiasts
| Content | Chemistry experiments - video | Physics experiments - video | Home Page - Chemistry and Chemists |
|
Chemistry and Chemists № 1 2026 Journal of Chemists-Enthusiasts |
Experiments with Universal Indicator - pt.12, 13 Chemist |
|
Having noticed a mistake in the text, allocate it and press Ctrl-Enter
Phenolphthalein Turns Fuchsia When Acid Is Added - Part 12
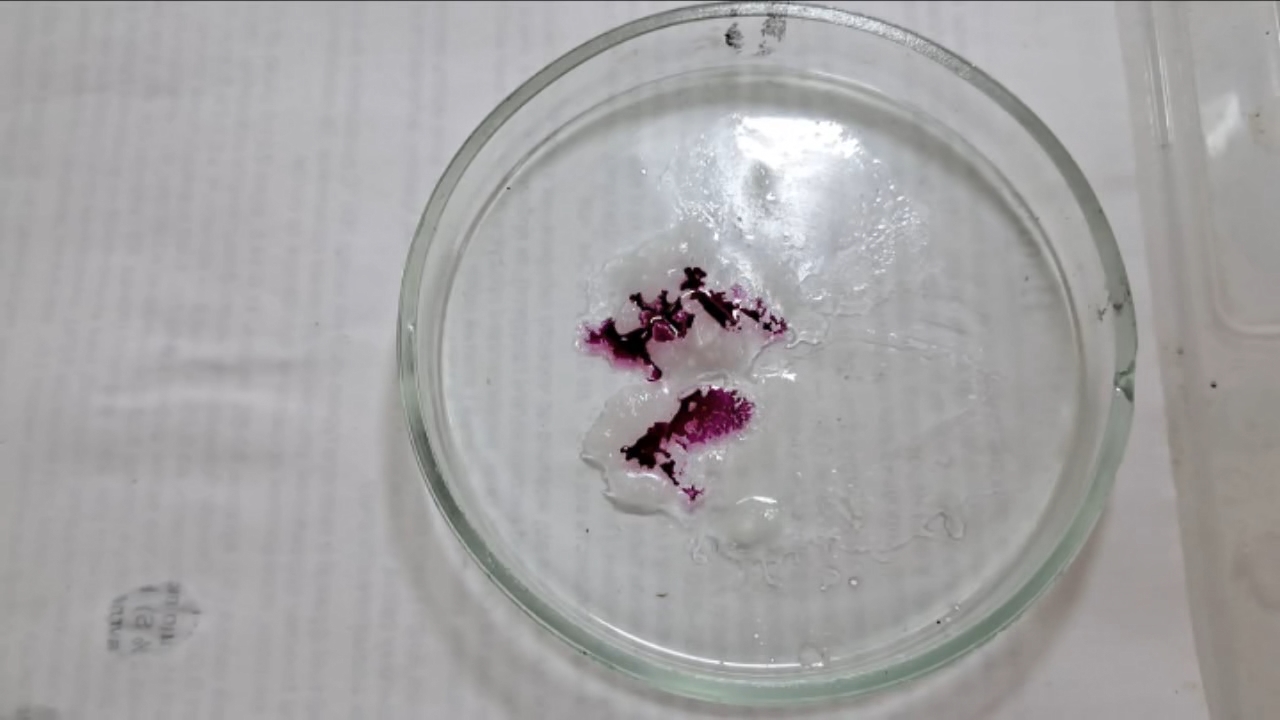
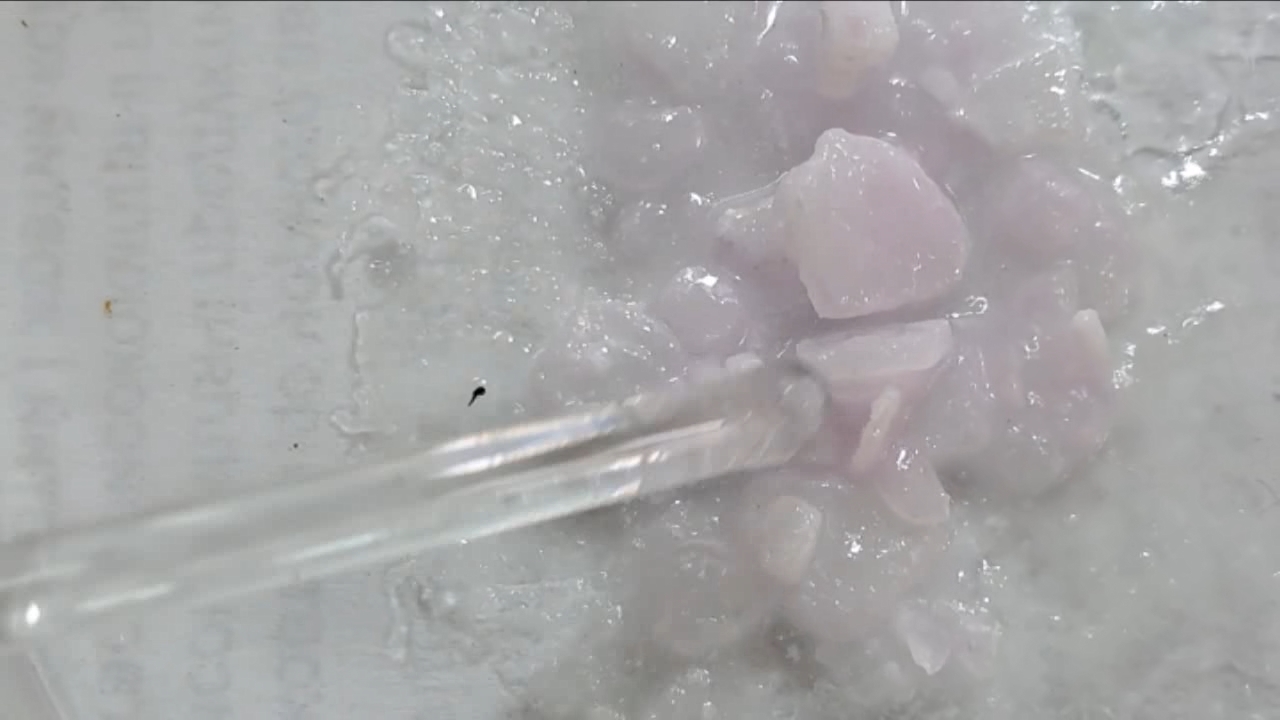
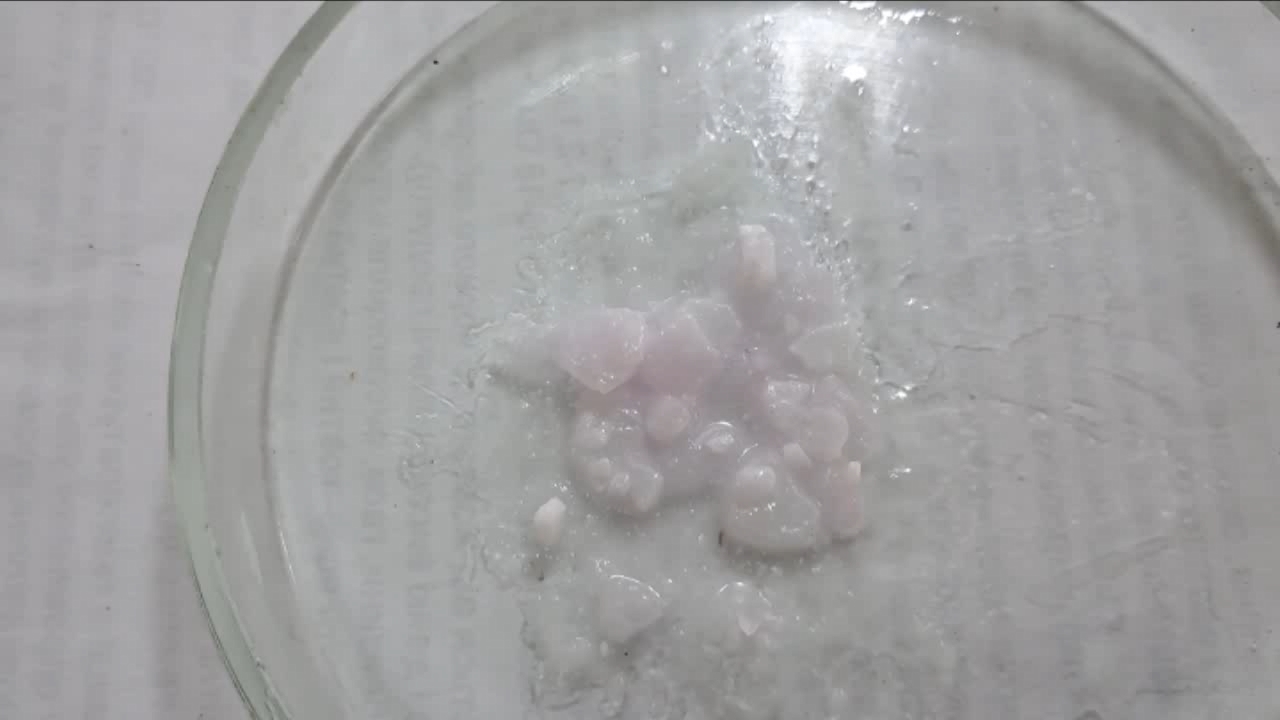
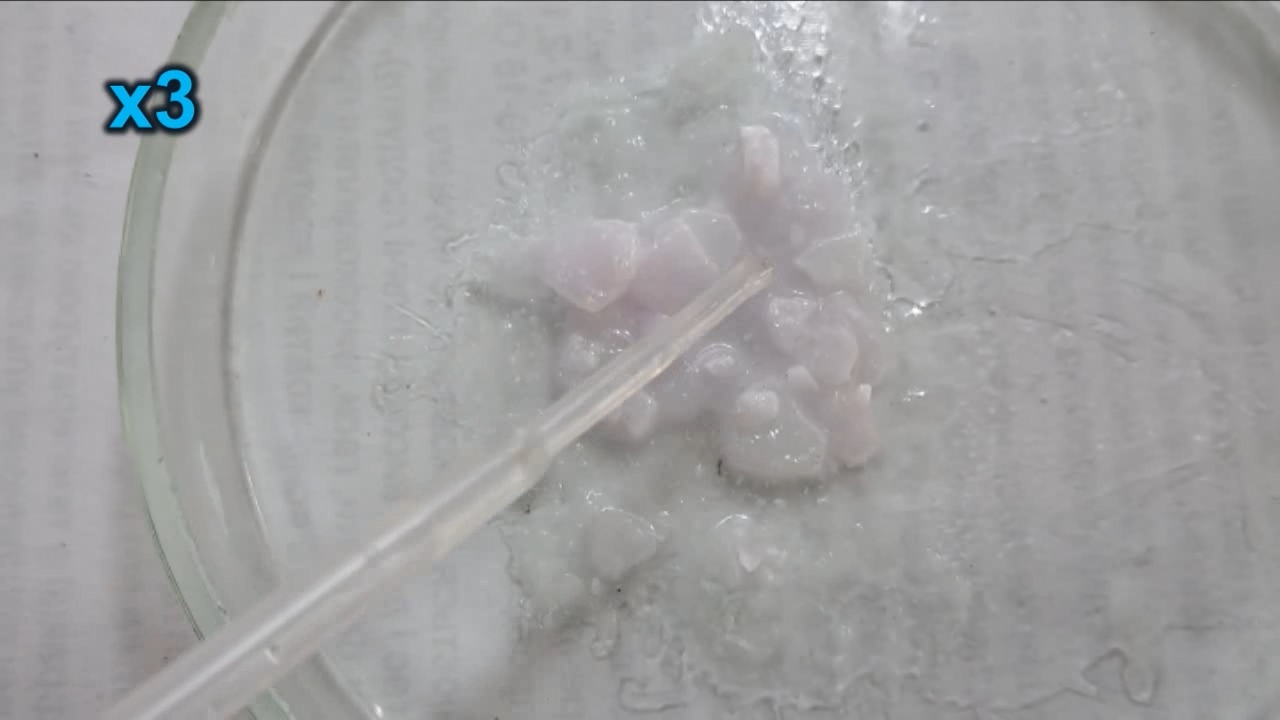
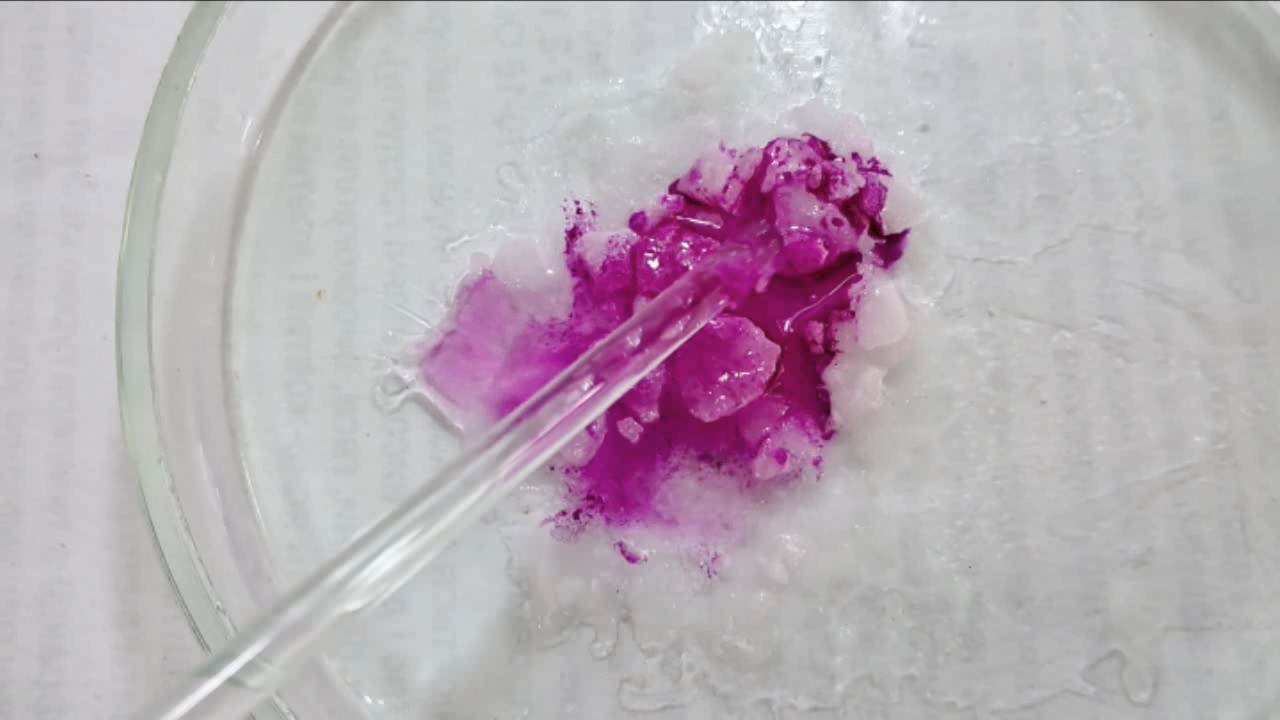
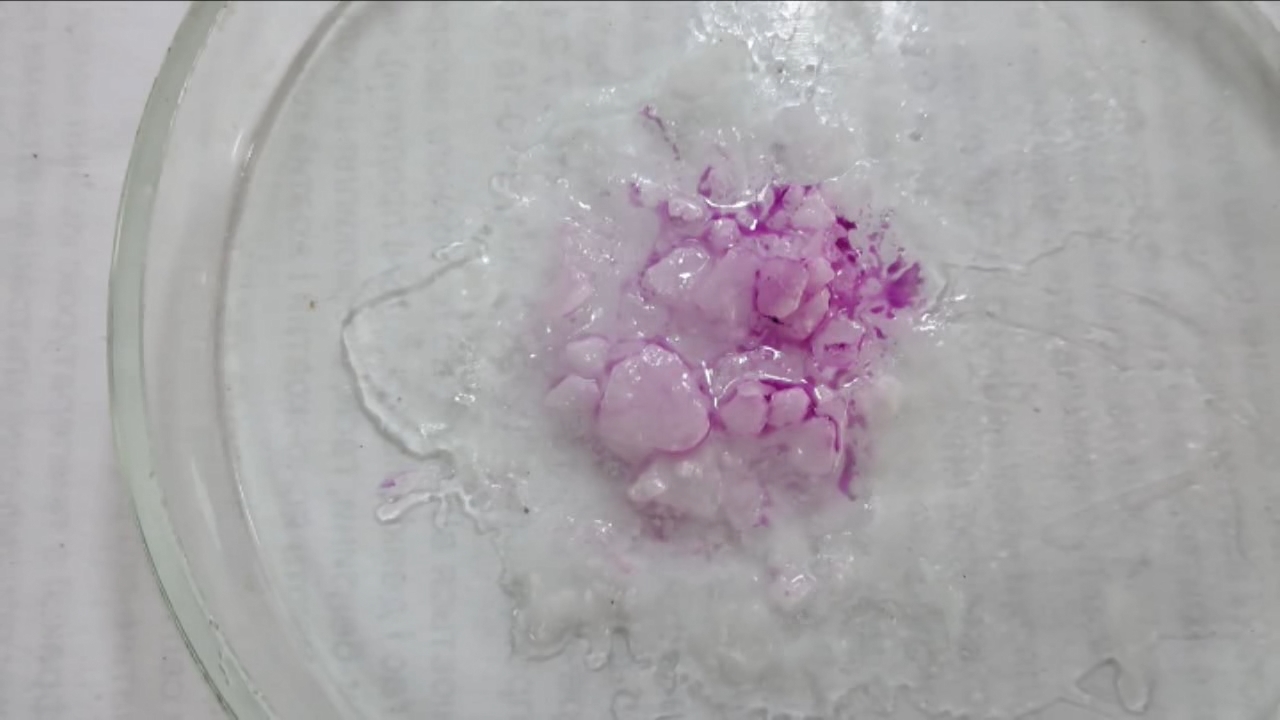
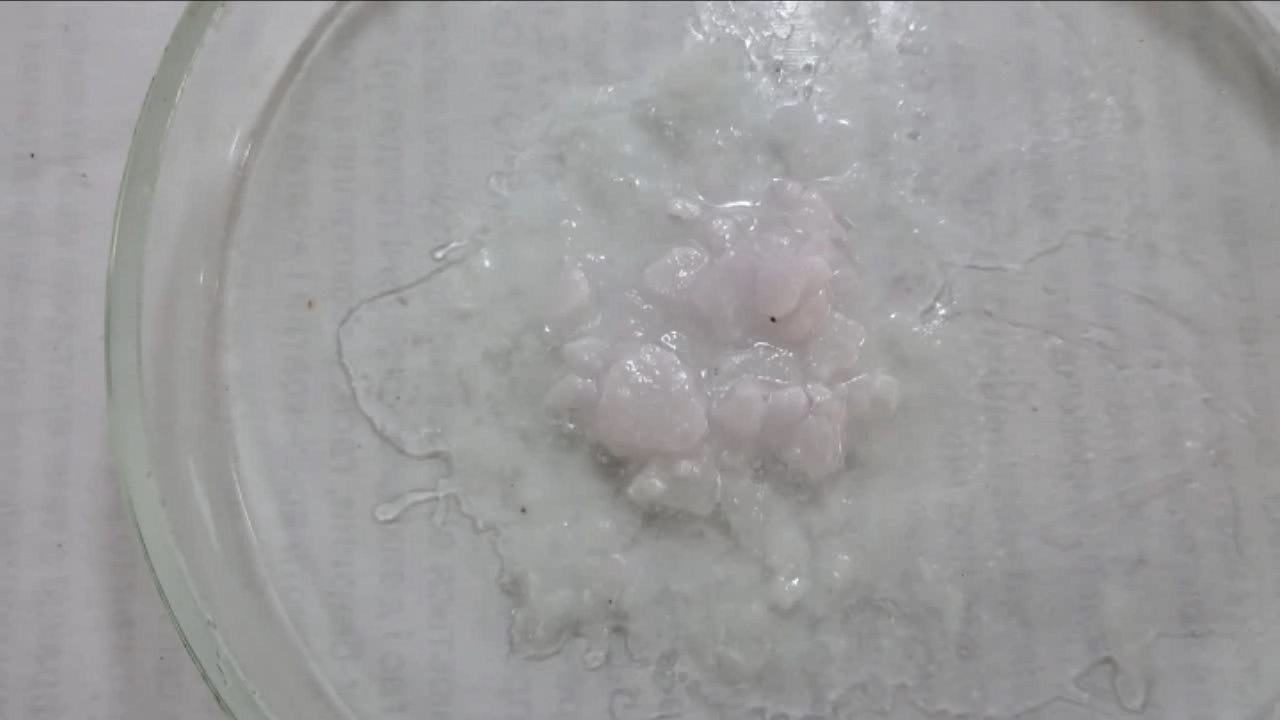
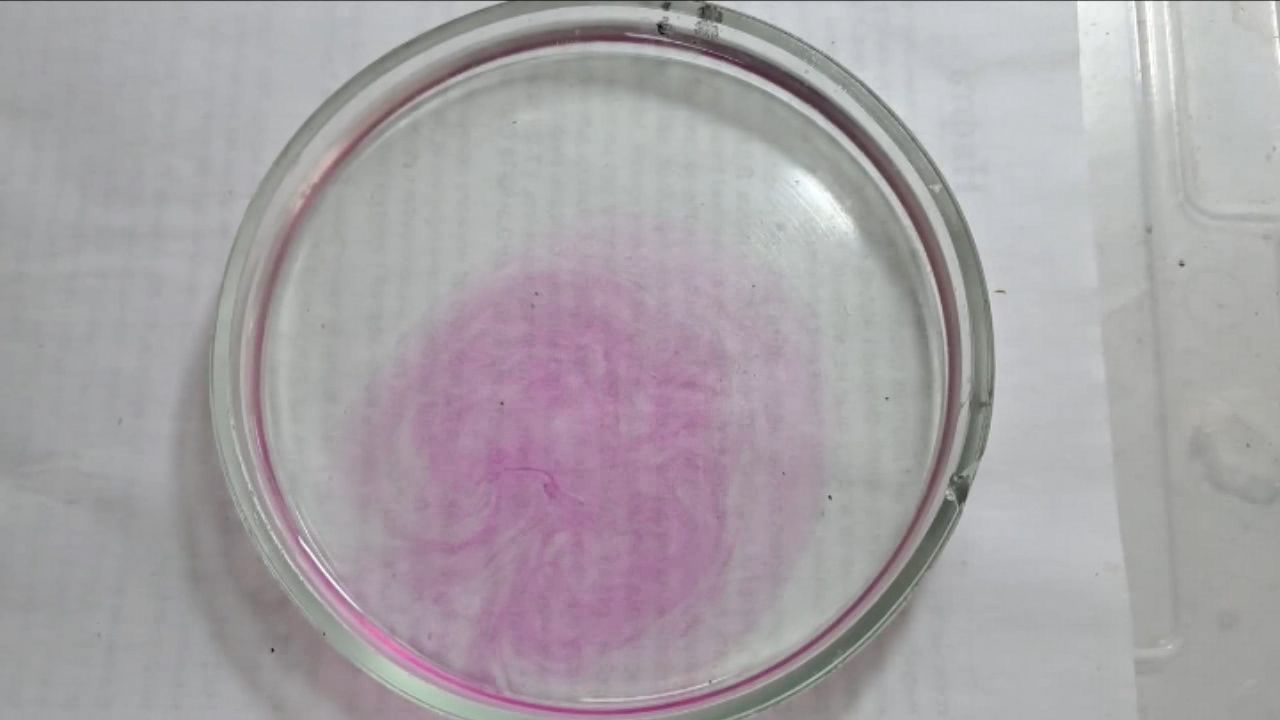
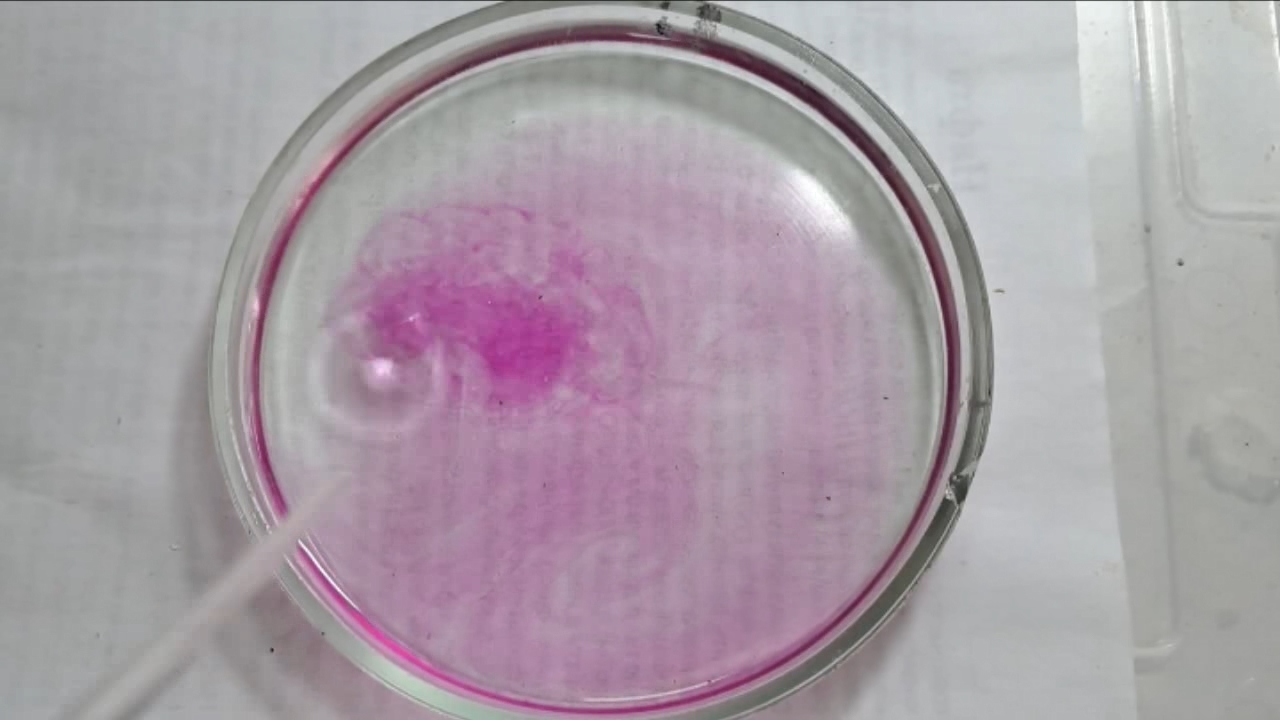
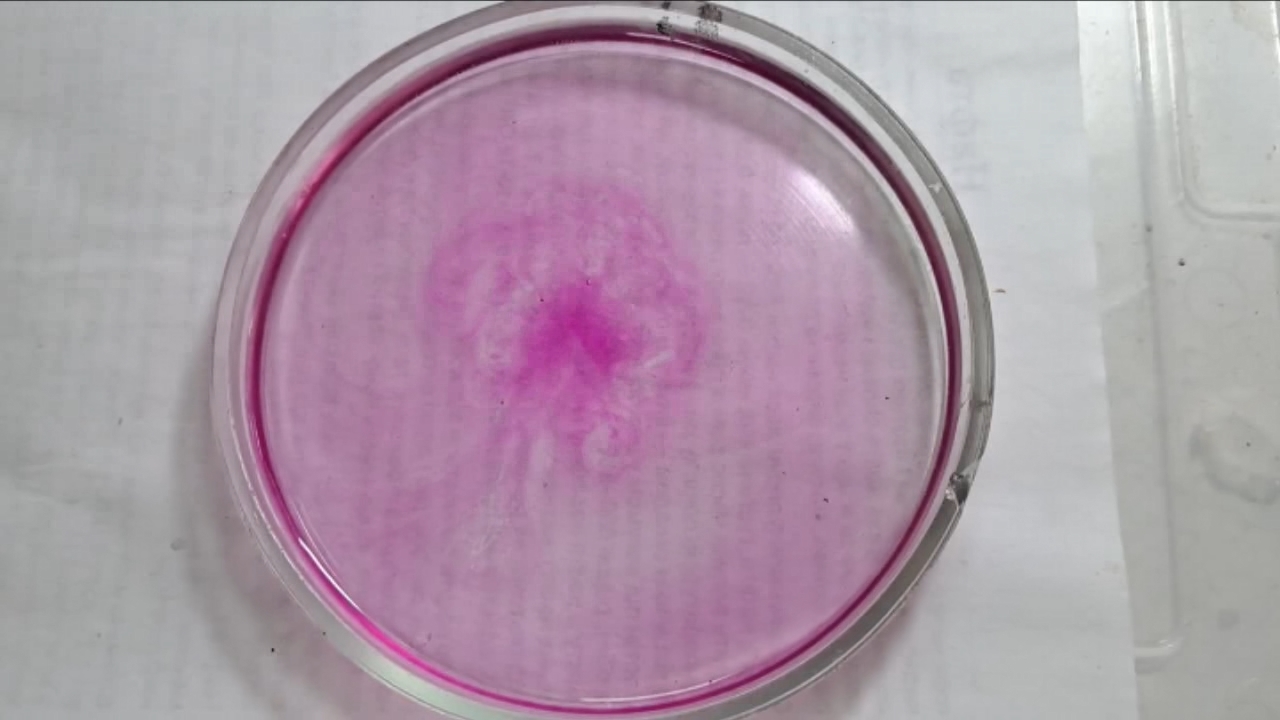
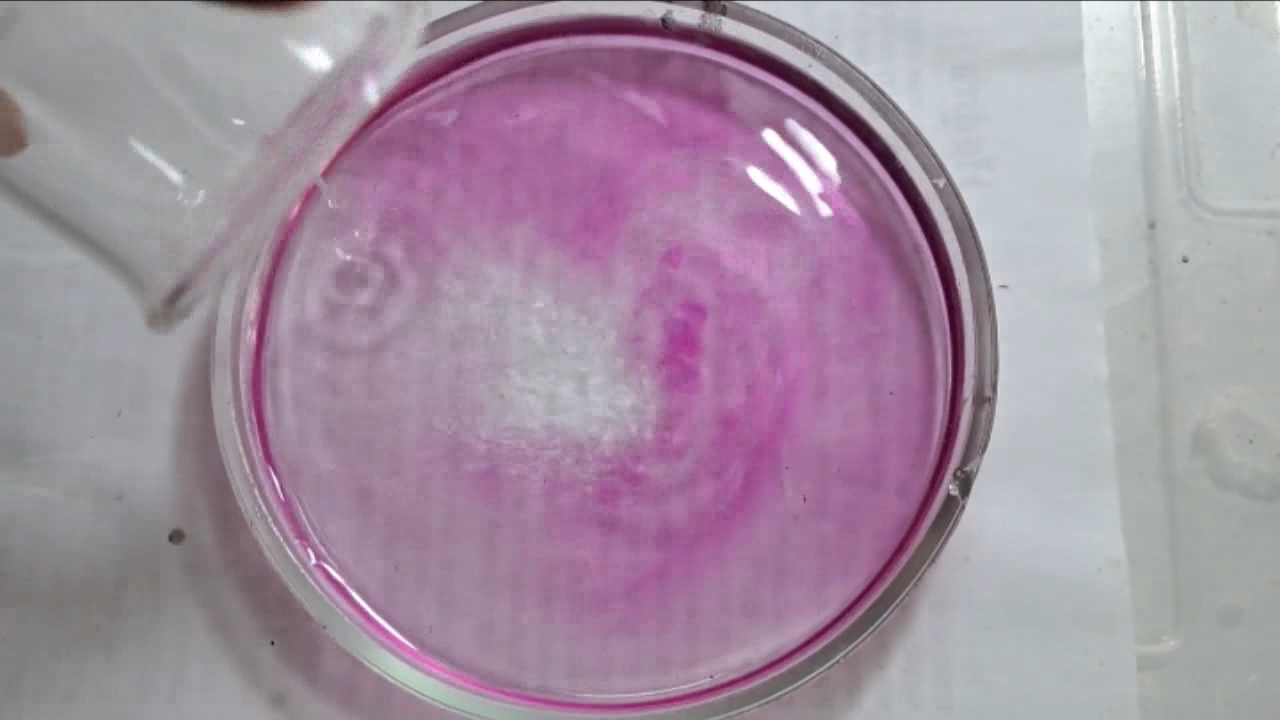
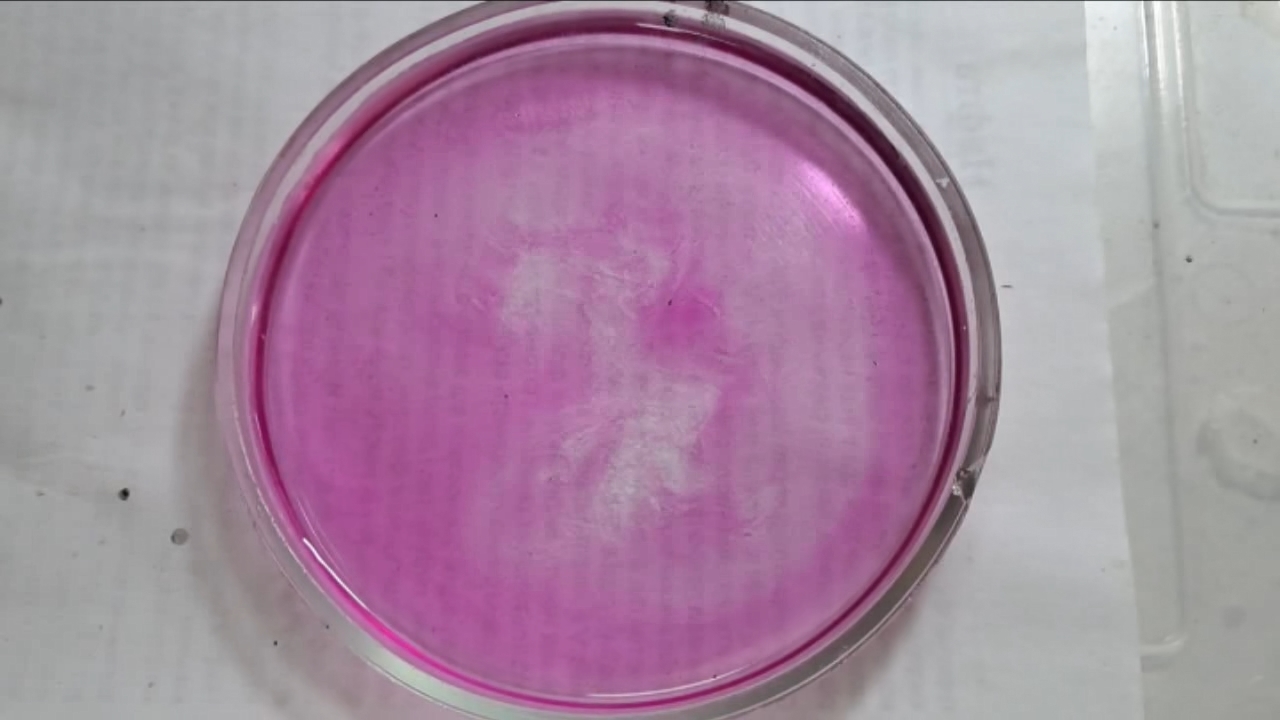
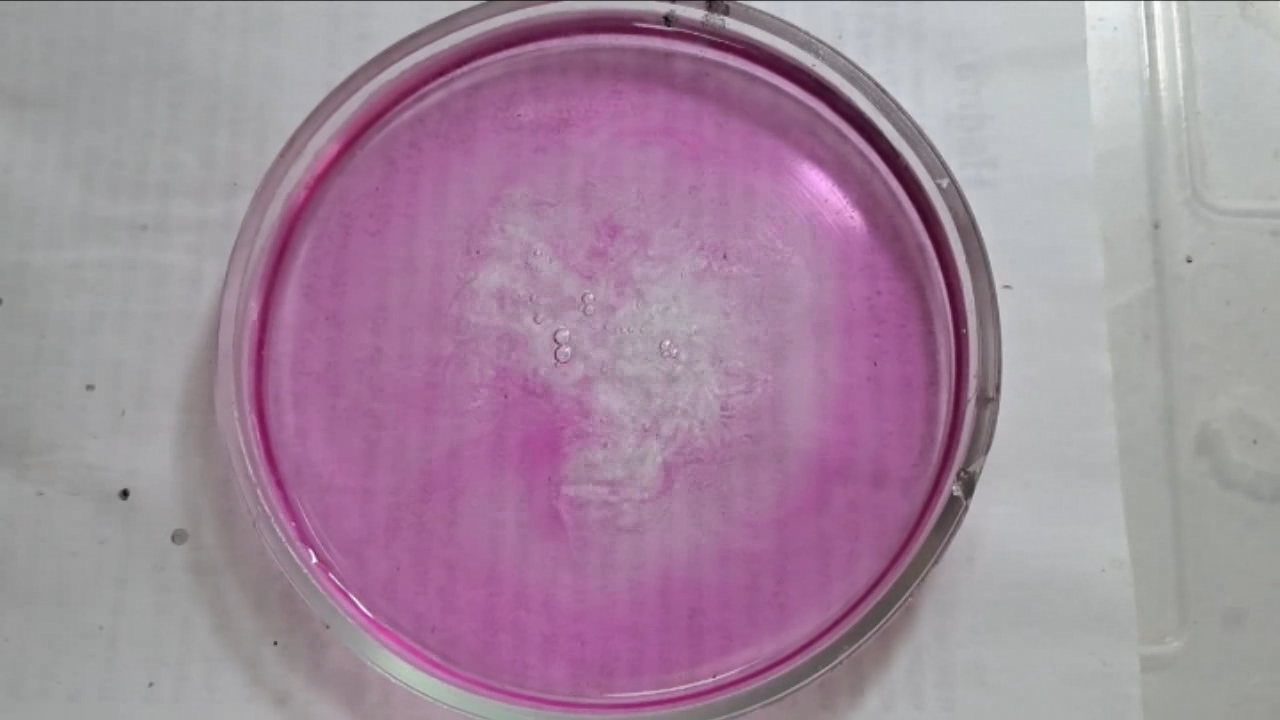
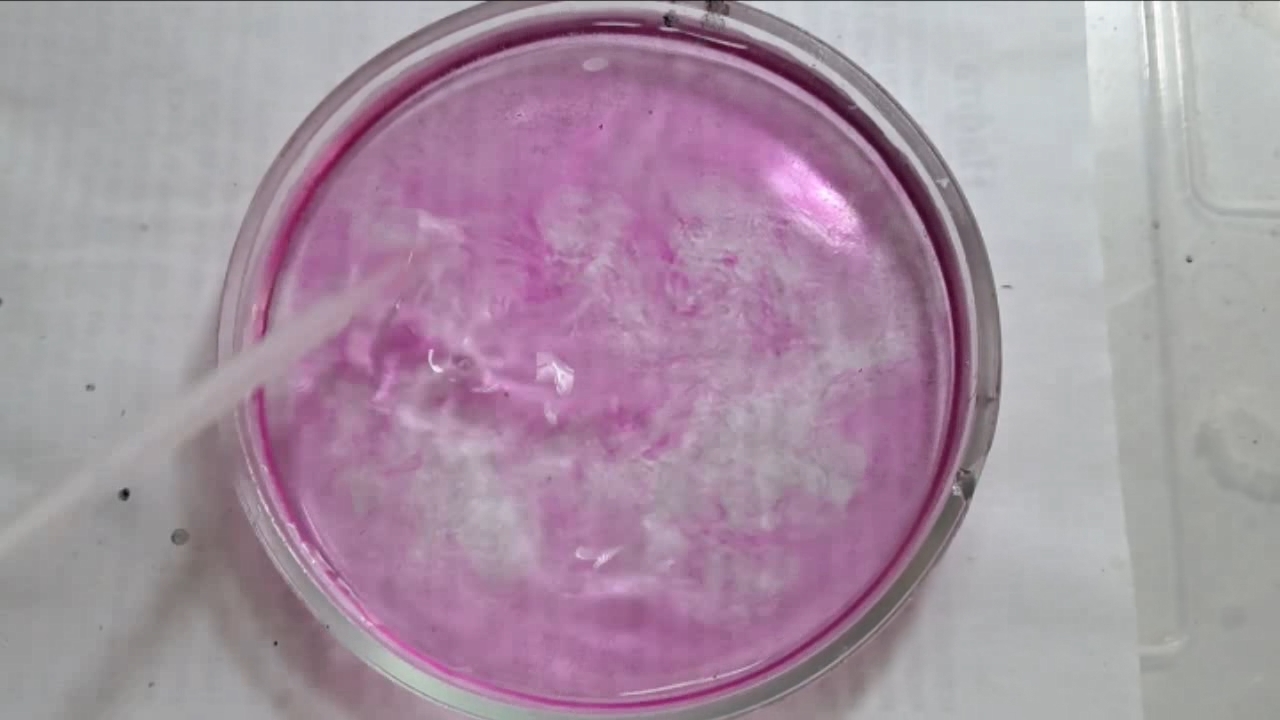
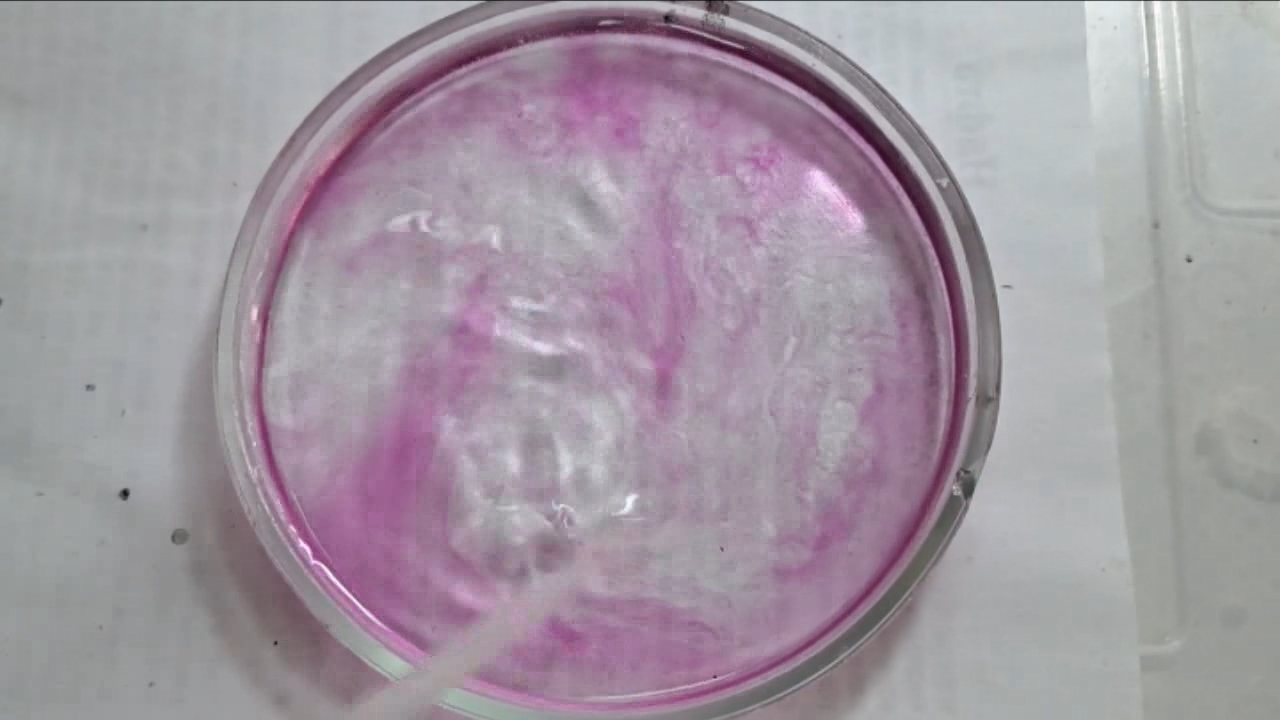
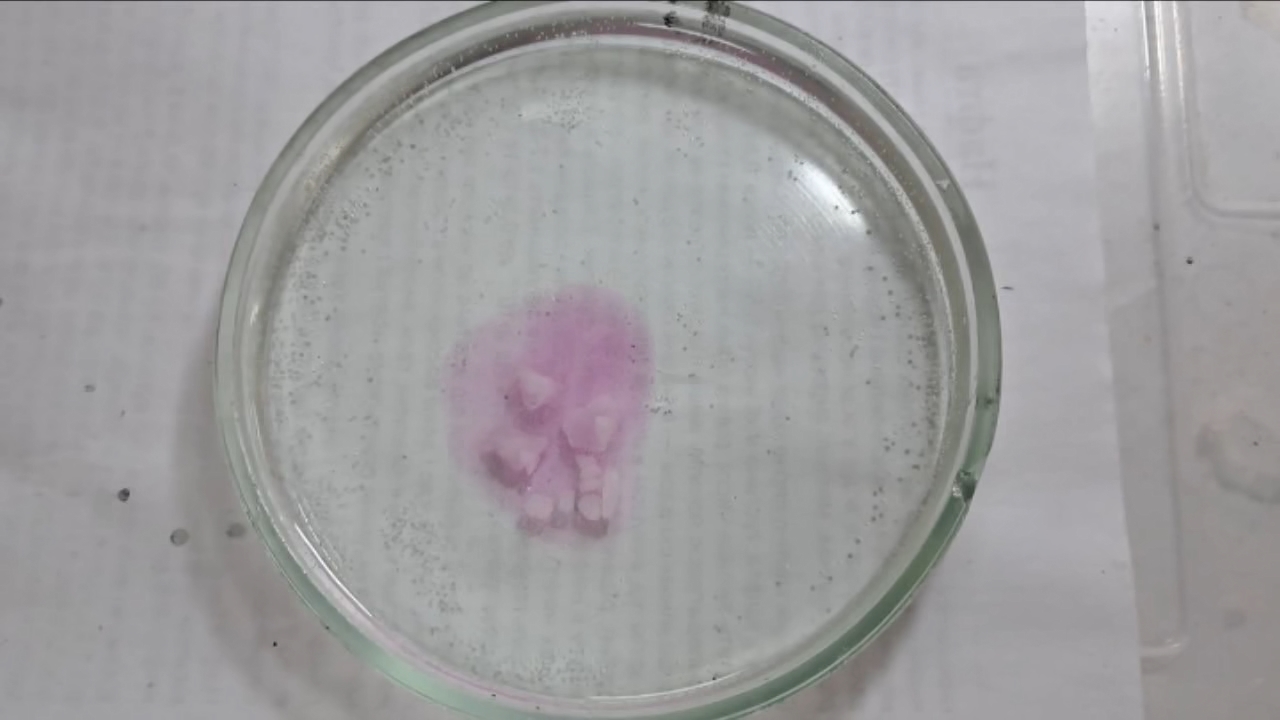
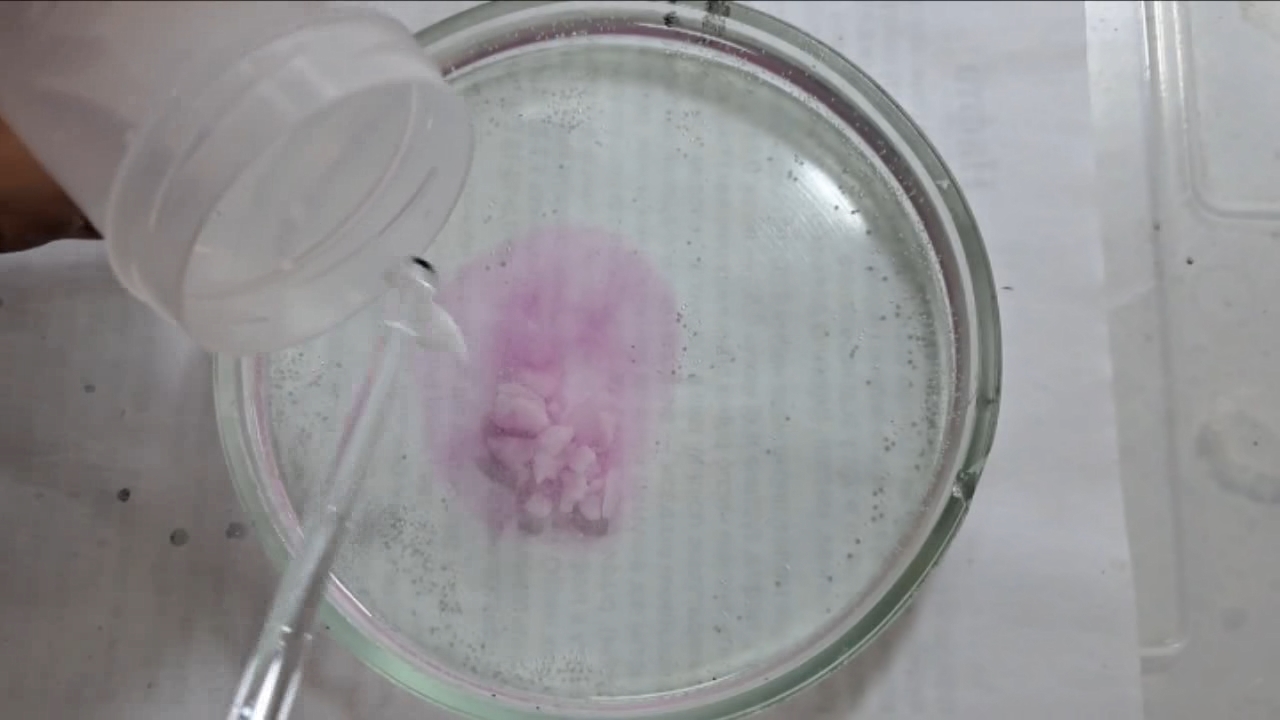
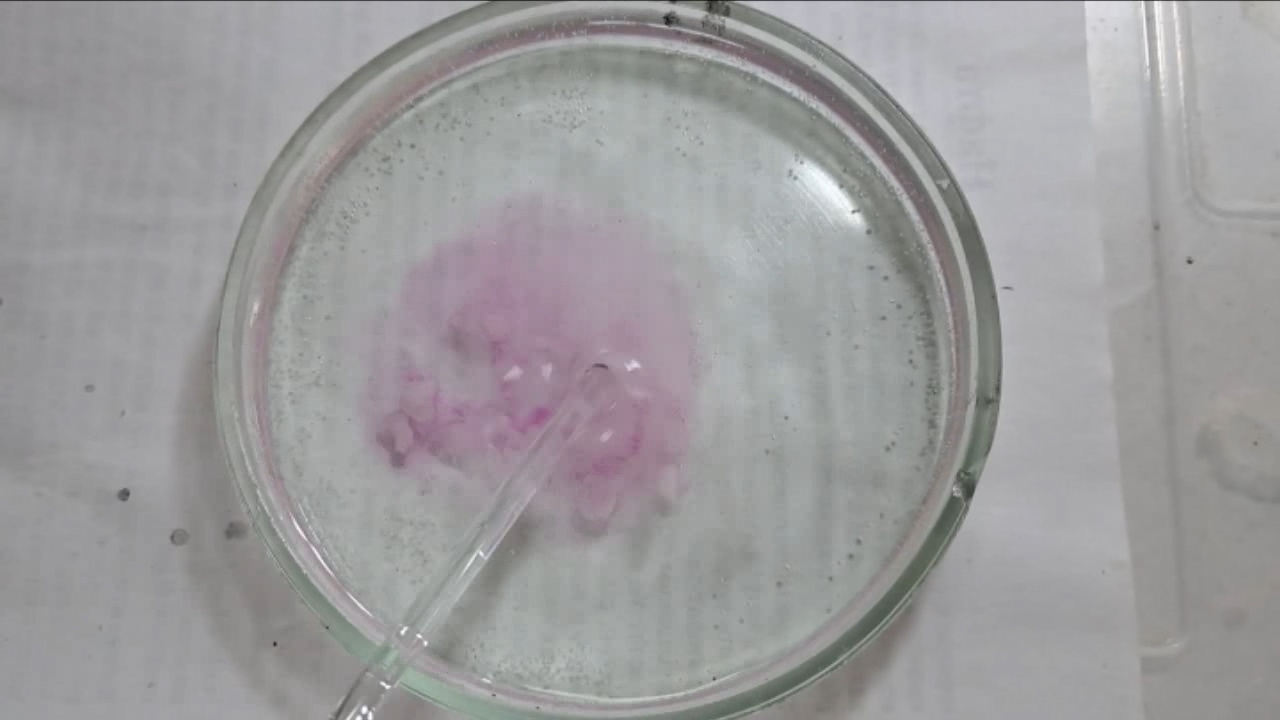
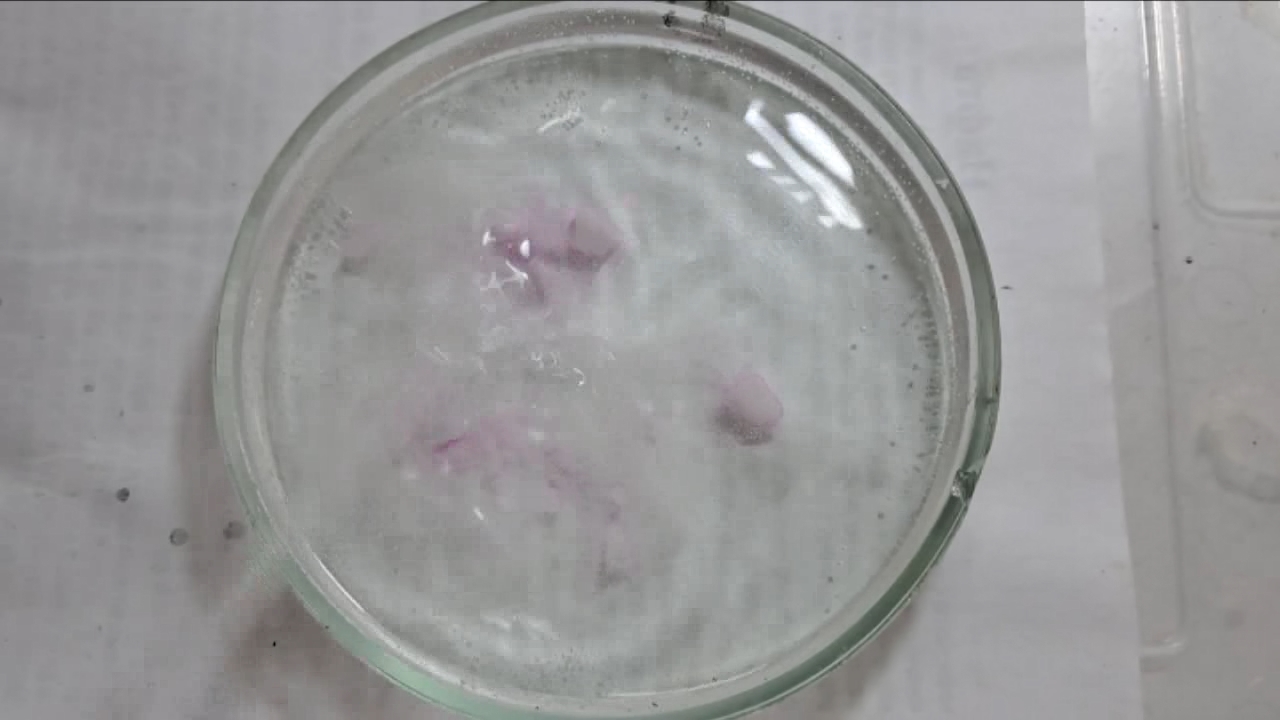
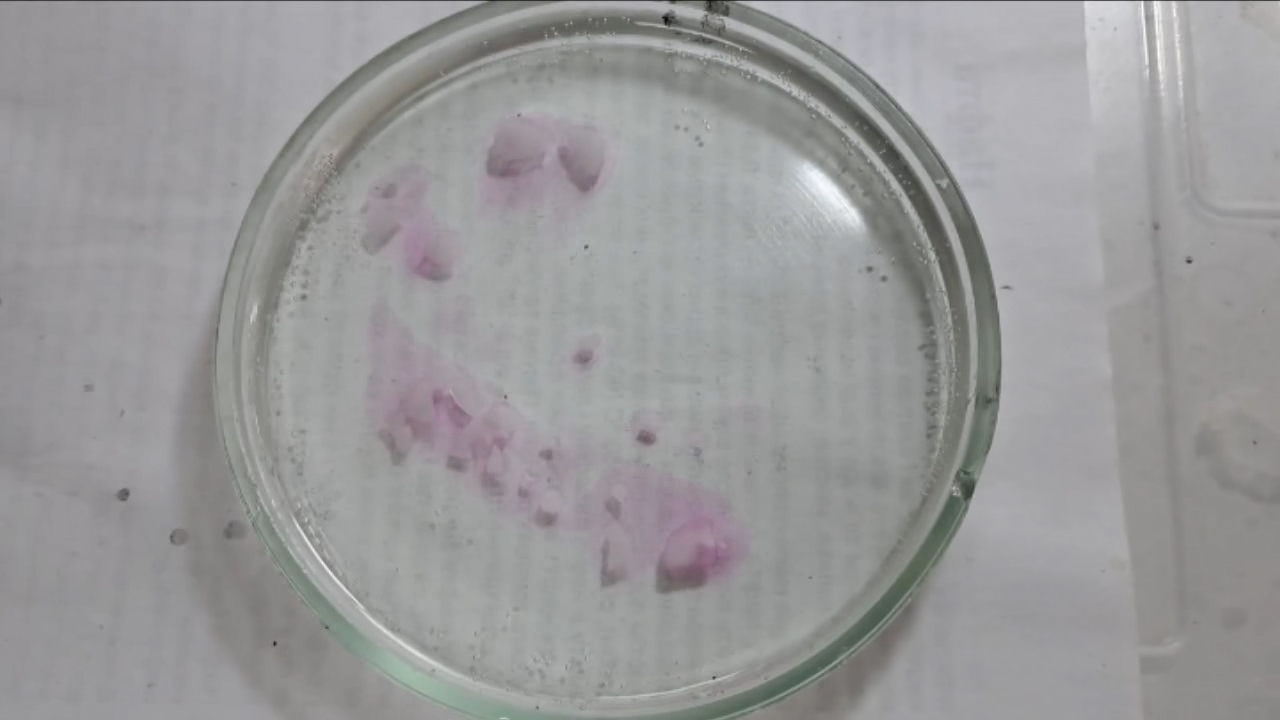
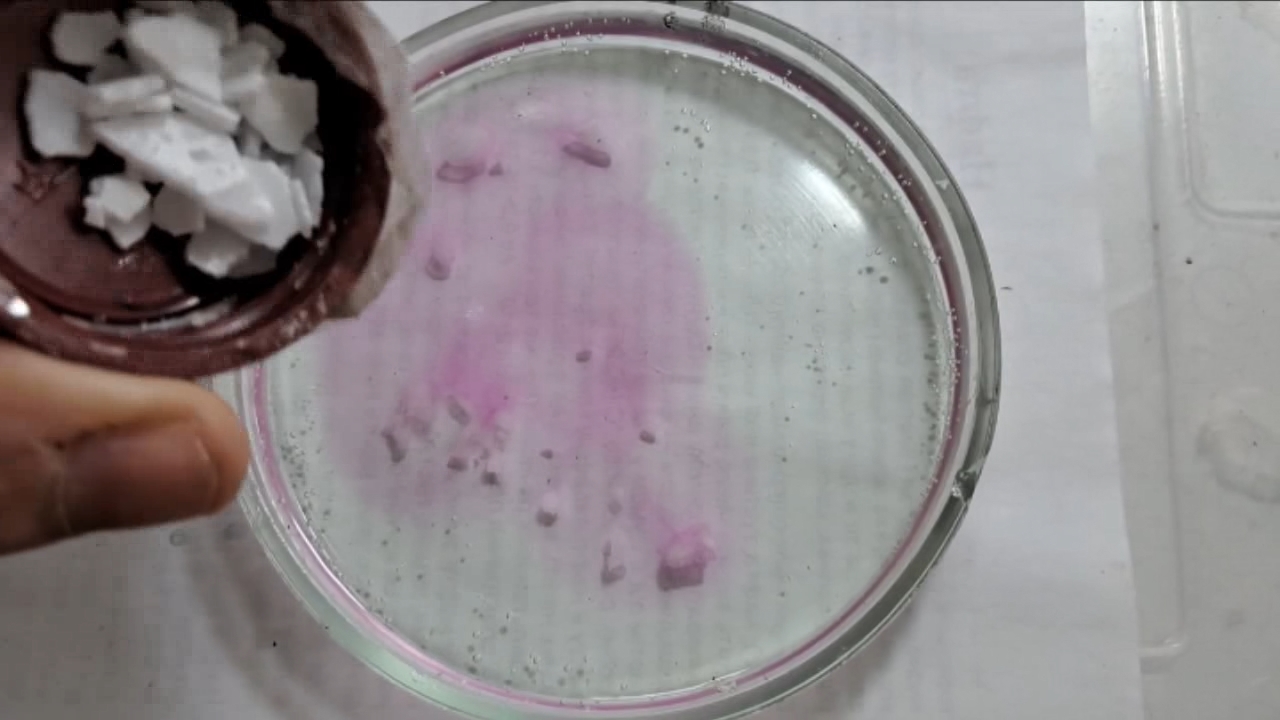
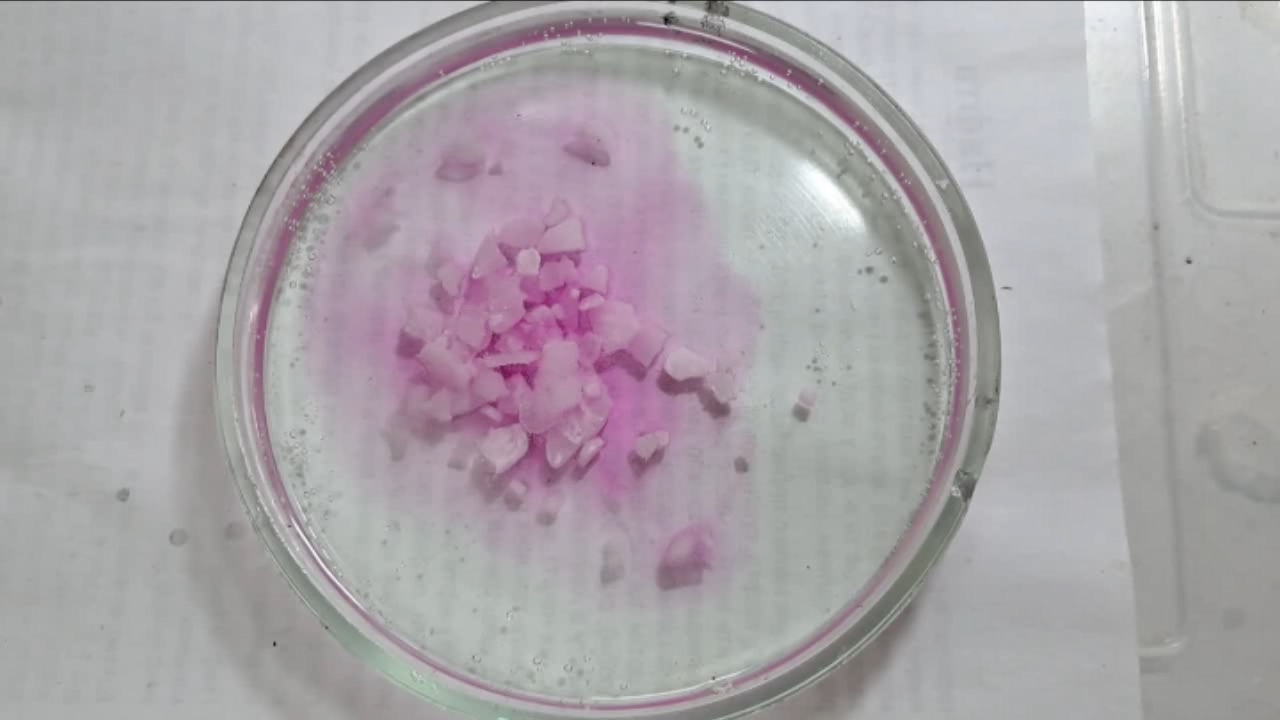
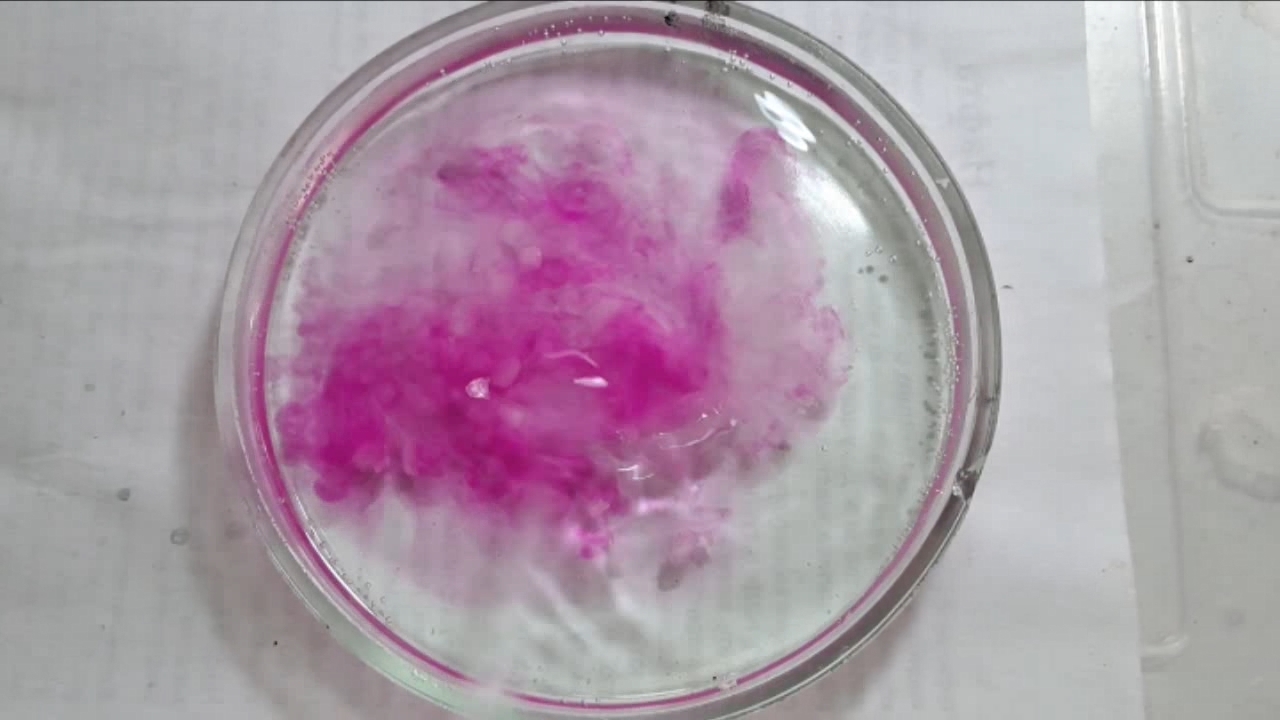
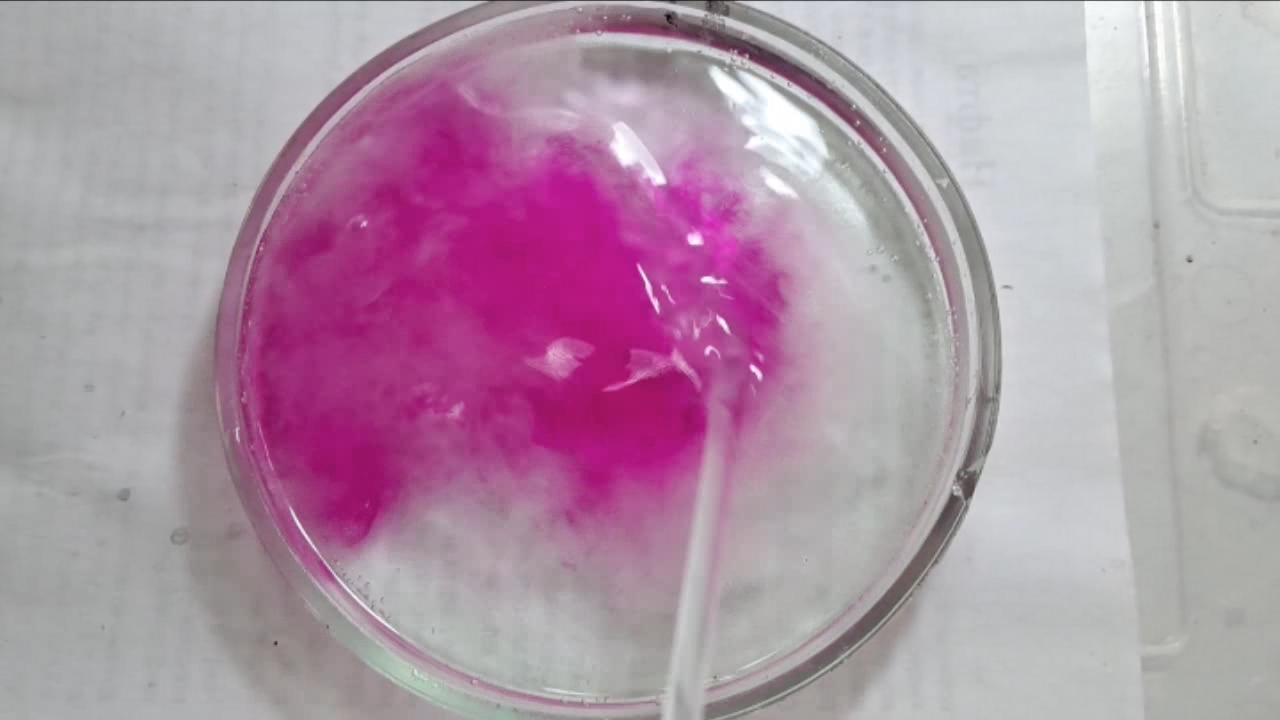
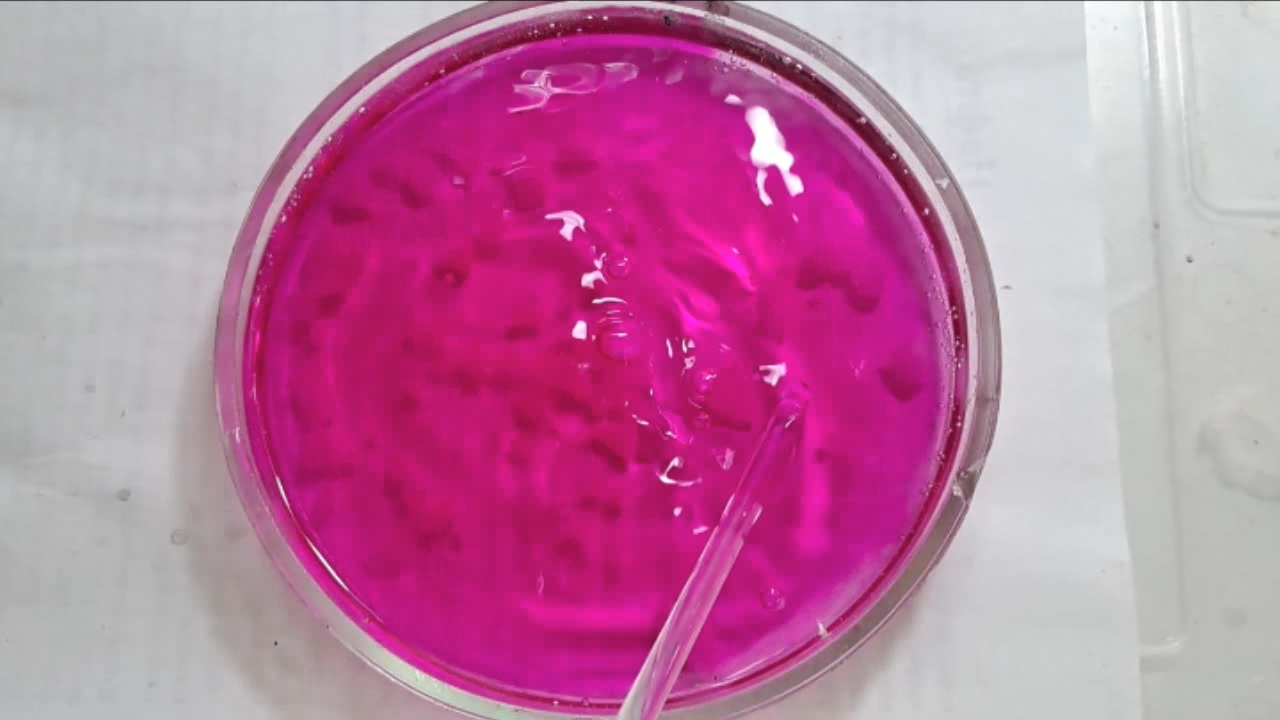
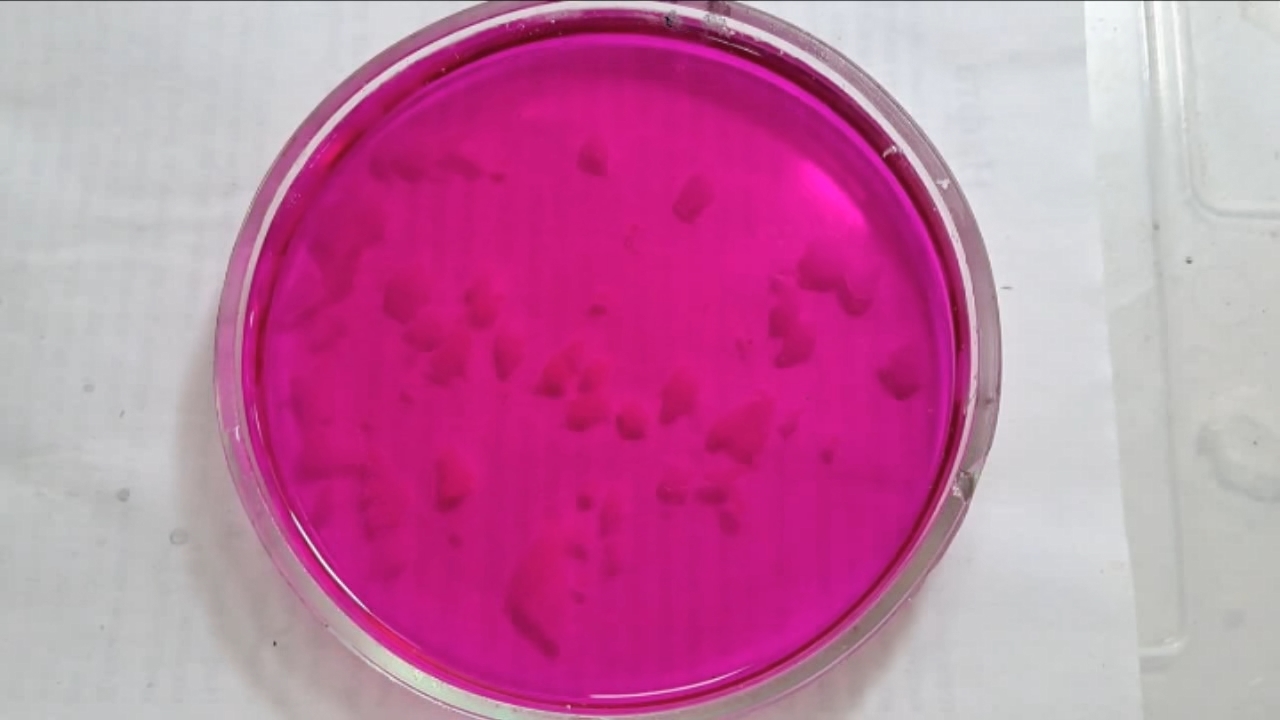
I placed a small pile of sodium hydroxide granules in a Petri dish and moistened them with water. I then added a few drops of an alcoholic solution of phenolphthalein. The mixture turned a deep fuchsia, almost black. After stirring the contents of the dish, the color faded somewhat but remained quite intense. I added additional sodium hydroxide granules and stirred again; the fuchsia color gradually began to fade.
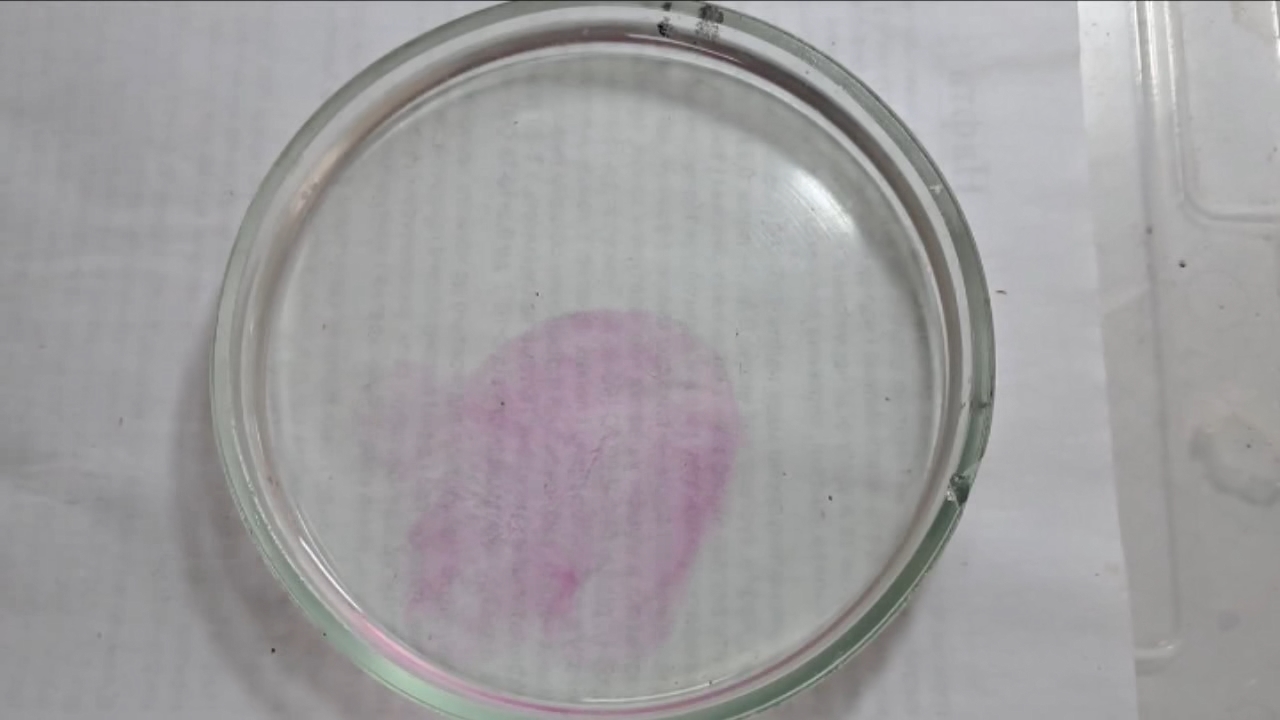
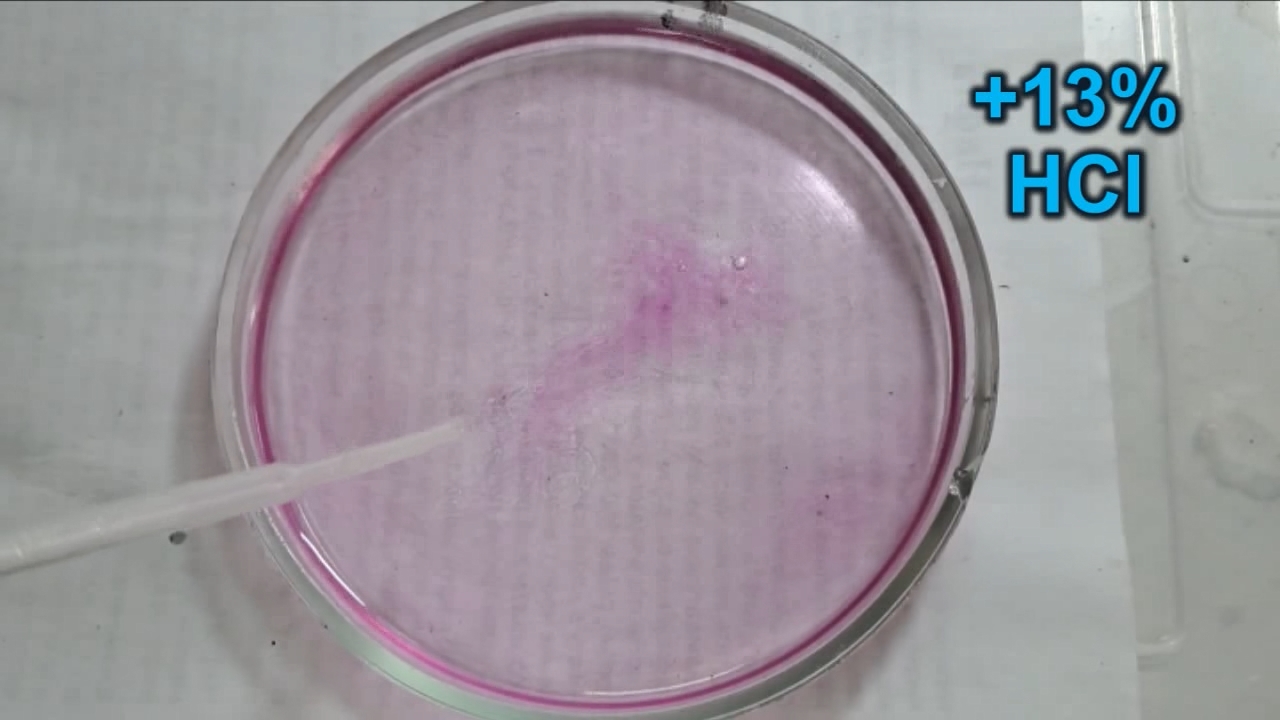
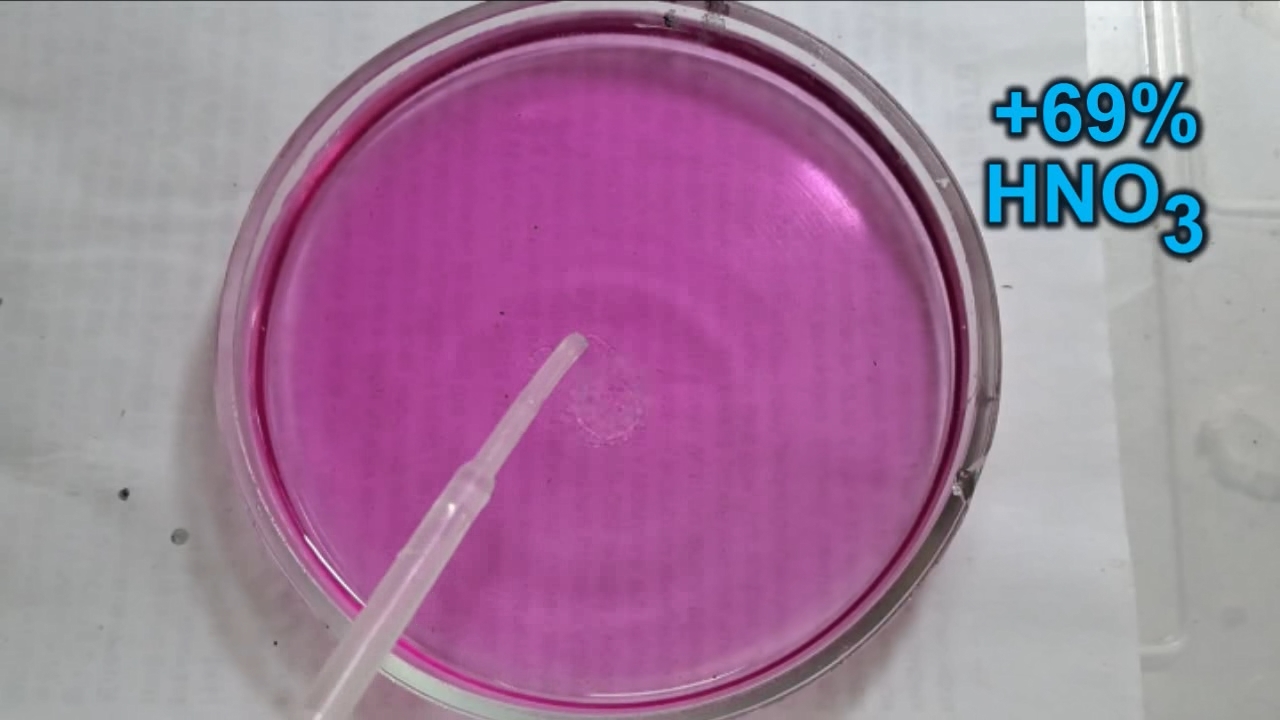
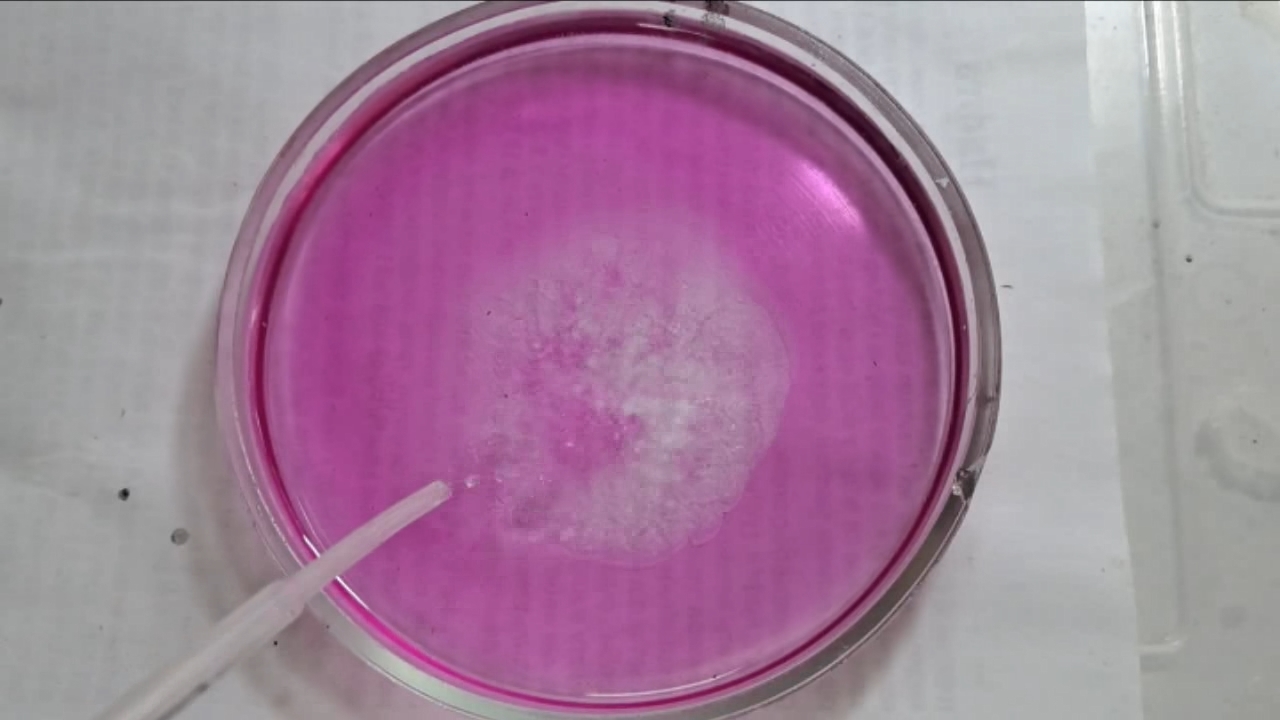
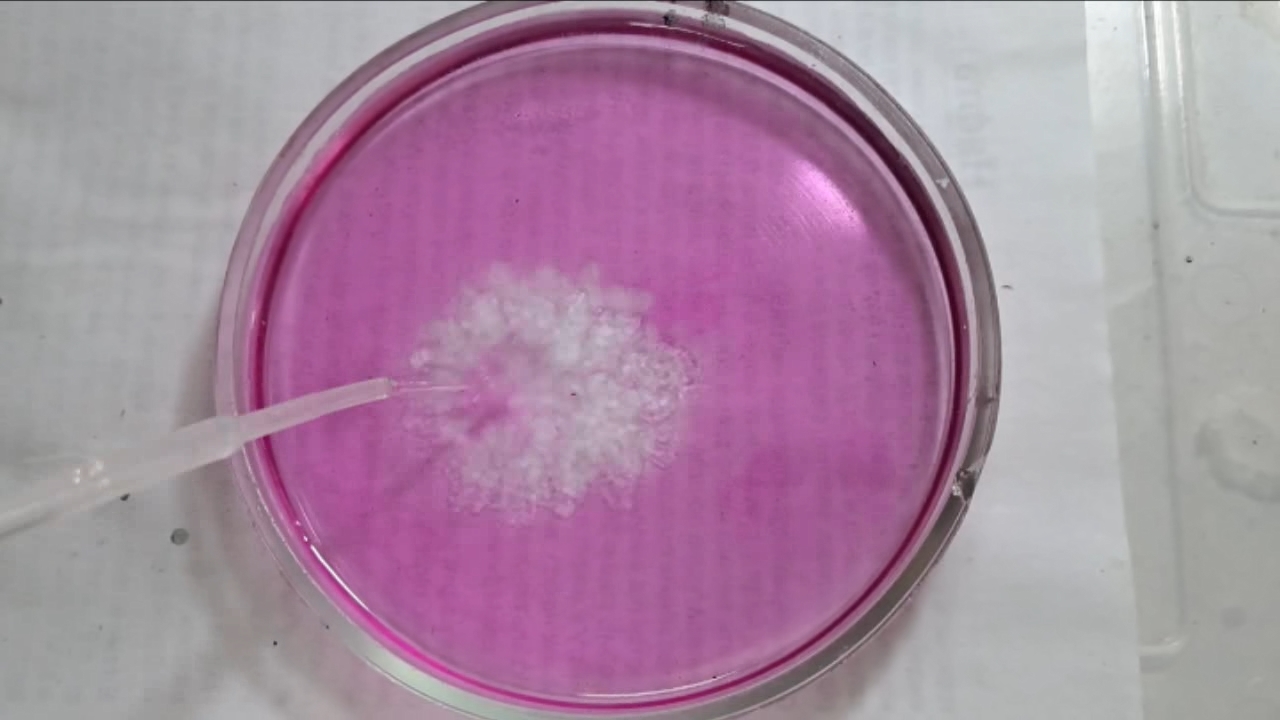
Фенолфталеин становится малиновым при добавлении кислоты - Часть 12 When the color had lightened to pale pink, I added a few more drops of phenolphthalein solution. The liquid again turned an intense fuchsia. After stirring, the color faded but remained quite bright. I realized a possible reason why the color faded only slowly despite a large excess of alkali. The sodium hydroxide was dissolved in water, whereas the phenolphthalein was dissolved in ethyl alcohol. The aqueous sodium hydroxide suspension and the alcohol were mixing slowly. I added a few drops of water and stirred; this accelerated the decolorization of the indicator. However, the process remained gradual. Even after several minutes, a faint pale pink tint persisted. I then added approximately 30 mL of water in order to reduce the sodium hydroxide concentration and induce the transition of phenolphthalein from its colorless form to the fuchsia one. I expected an immediate appearance of intense coloration. Instead, the opposite occurred: the liquid remained colorless, and the residual pink tint disappeared. Apparently, the alkali concentration was still sufficiently high to keep the phenolphthalein in its colorless form. To reduce the sodium hydroxide concentration further, an acid had to be added. I first introduced a small amount of solid glacial acetic acid; the crystals dissolved without producing any visible coloration. I then began adding a 13% hydrochloric acid solution dropwise. The very first drop caused a fuchsia spot to appear. With subsequent additions, the spot expanded and intensified in color. Eventually, the entire solution turned pink. After adding more acid and stirring, the liquid in the Petri dish became bright fuchsia. Thus, upon addition of hydrochloric acid, the phenolphthalein turned fuchsia! I then added 69% nitric acid dropwise in order to neutralize the alkali and cause the indicator's color to disappear. The first few drops produced no decolorization. I added several larger portions (approximately 0.5 mL each). Each addition caused local decolorization, but the fuchsia color quickly returned. The acid was clearly in excess, yet the solution in the dish remained intensely colored. Why did the phenolphthalein not decolorize after the addition of an excess of nitric acid? The explanation proved to be simple. The nitric acid solution, with a density of about 1.4 g/mL, sank to the bottom and mixed only slowly with the less dense alkaline solution. Once the liquid was stirred, the phenolphthalein rapidly became colorless. I then added a few sodium hydroxide granules. Carbon dioxide began to evolve, and the liquid surrounding the granules turned fuchsia. Upon stirring with a glass rod, the colored regions nearly disappeared, but then reappeared as the solid alkali continued to dissolve. After adding an excess of sodium hydroxide and stirring thoroughly, the acid was neutralized. The liquid in the dish became bright fuchsia. Finally, it should be emphasized that splashing may occur when mixing concentrated acids and alkalis. Protective goggles, gloves, and appropriate clothing should always be worn. Contact with strong alkali can cause severe and irreversible eye damage. |

Phenolphthalein Turns Fuchsia When Acid Is Added |

|
|

|

|

|

|

|
|
|

|
|

|

|

|
|
|
|

|

|

|

|

|
|
|
|
|
|
|

|
|
|

|

|
|
|
|

|
|
|

|

|

|

|
|
|
|
|
|
|

|
|
|
|
|
|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|