Chemistry and Chemists № 1 2026
Journal of Chemists-Enthusiasts
| Content | Chemistry experiments - video | Physics experiments - video | Home Page - Chemistry and Chemists |
|
Chemistry and Chemists № 1 2026 Journal of Chemists-Enthusiasts |
Experiments with Universal Indicator - pt.1, 2, 3 Chemist |
|
Having noticed a mistake in the text, allocate it and press Ctrl-Enter
What Is a Universal Indicator? (Introduction) - Part 1
pH indicators (acid-base indicators) are organic dyes that change color depending on the hydrogen ion concentration in a solution. The color transition of an indicator occurs over a specific pH range. Some indicators may exhibit two or more such ranges.
Эксперименты с универсальным pH индикатором Что такое универсальный индикатор? (Вступление) - Часть 1 A universal indicator is a mixture of several organic dyes, each of which changes color over a different pH range. Unlike individual acid-base indicators, a universal indicator changes color over a wide pH range (e.g., from 1 to 12 or from 1 to 14), allowing not only the qualitative detection of acids or alkalis in a solution but also an approximate determination of hydrogen ion concentration by comparing the indicator color with a standard scale. For example, a universal indicator may contain thymol blue, methyl red, methyl orange, bromothymol blue, and phenolphthalein. The indicator can be used in the form of paper strips (universal indicator paper) or as a solution. 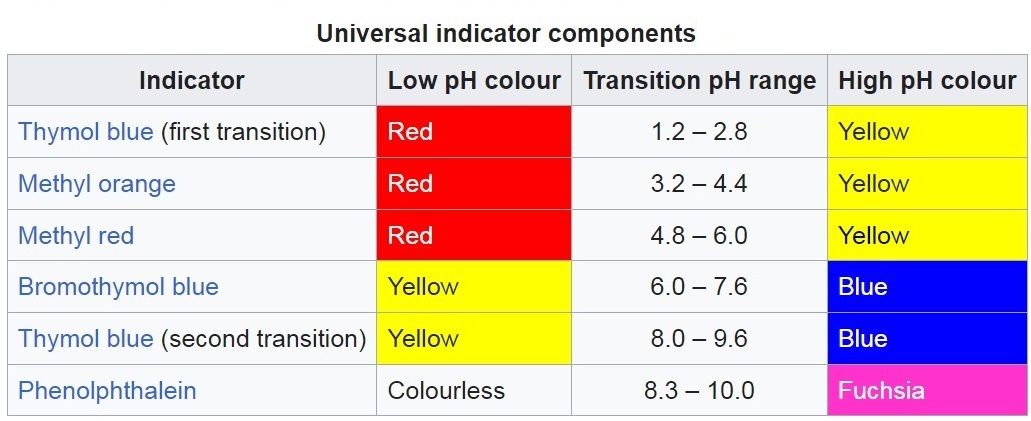
 
|

Universal indicator paper |

|

|
|

|

|

|

Litmus paper |

|

|
|

|

|

|

|

|
|
Having noticed a mistake in the text, allocate it and press Ctrl-Enter
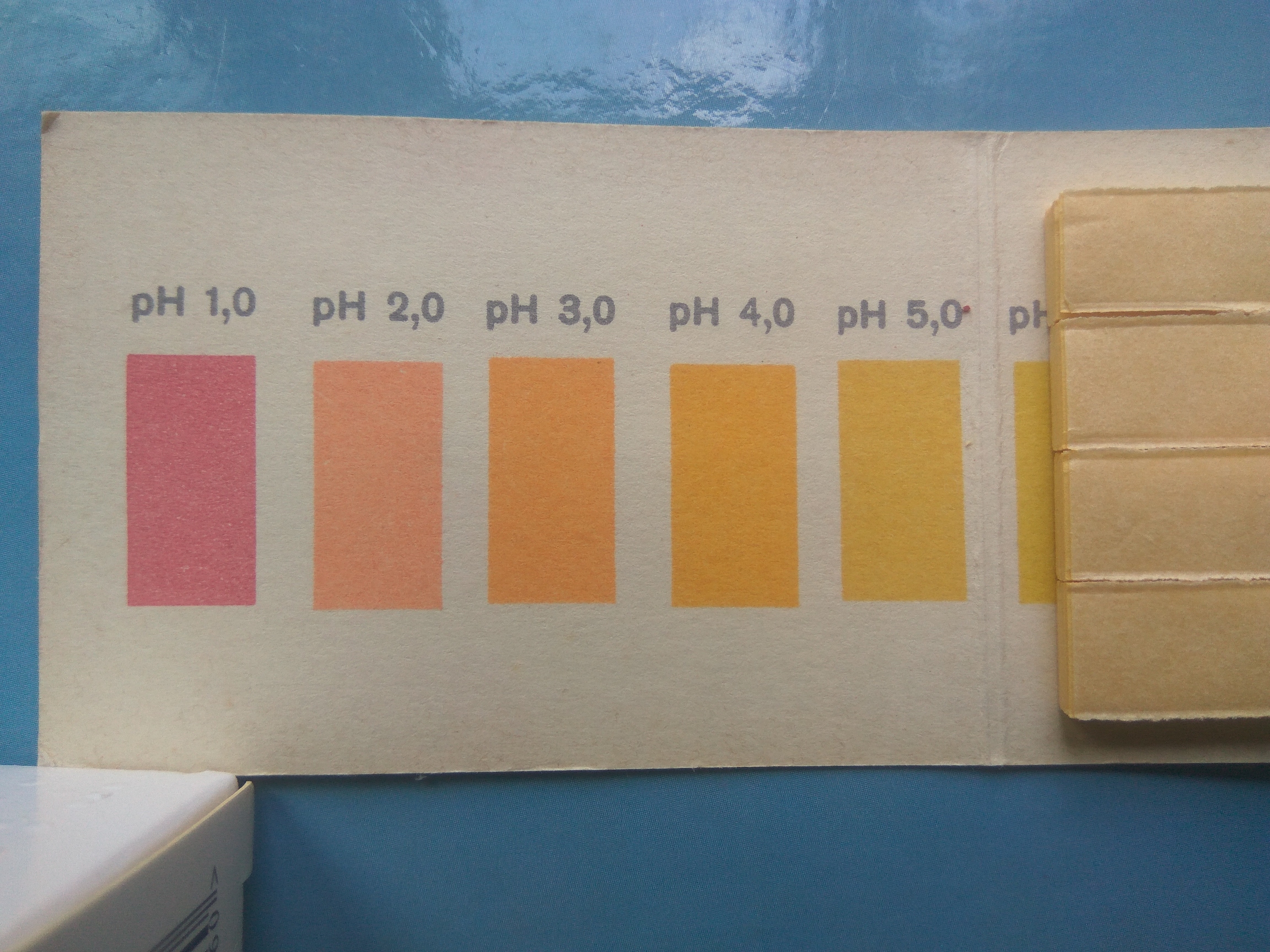
As is well known, pure water has a pH of 7. Acidic solutions have a pH< 7, while alkaline solutions have a pH >7. In the former case, the hydrogen ion concentration is higher than in pure water, while in the latter, it is lower. If the pH of one solution is one unit lower than that of another, then the hydrogen ion concentration in the first solution is ten times higher than in the second.
Perfectly pure water has a neutral pH, exactly 7, but nature does not favor perfection. For example, tap water is usually slightly alkaline due to the hydrolysis of dissolved salts. Therefore, its pH is typically slightly above 7. Distilled water contains virtually no dissolved salts, but it usually contains dissolved carbon dioxide, which enters the water from the air. Carbon dioxide reacts with water to form weak carbonic acid; therefore, distilled water often has a pH below 7. For example, when I measured the pH of distilled water, I obtained values of 6.5, 6.3, or even as low as 5.5. To remove carbon dioxide from water, it can be boiled for an extended period and then cooled in a sealed flask connected to the atmosphere via a tube filled with barium hydroxide. The indicator phenolphthalein has a color transition at approximately pH 8.2. At pH < 8.2, phenolphthalein is colorless (in acidic, neutral, and slightly alkaline solutions), whereas at pH ≥ 8.2 it develops a crimson (pink-fuchsia) color [1]. If tap water is poured into one glass and distilled water into another, and a few drops of phenolphthalein solution are added to both, the indicator remains colorless in each case. If the glasses are then heated on an electric hotplate, the distilled water remains colorless, whereas the tap water soon acquires a faint pink color that intensifies upon heating. At boiling, the solution becomes intensely fuchsia. Heating enhances the hydrolysis of salts and also causes the decomposition of bicarbonates to form carbonates, which increases the pH, making the solution more alkaline. As a result, the phenolphthalein transition point is reached, and the solution turns fuchsia. After cooling, the reverse process begins, but it does not occur instantly. In my experiment [2], the phenolphthalein color faded very slowly. Even after three days, a faint pink tint was still visible. I decided to repeat this experiment using a universal pH indicator instead of phenolphthalein. A brief digression. I have been working in chemistry for about 38 years. Since childhood, I have used universal indicator paper - exactly the type shown in the photographs in Part 1. However, I have encountered a universal indicator in powdered form in a bottle only a few times. Although the components of a universal indicator are relatively inexpensive and readily available, it is most commonly sold as indicator paper. Last year, a colleague gave me a bottle of this universal indicator. I put it in my desk drawer to avoid losing it and planned to experiment with it. However, due to the winter cold, I had to move to another laboratory, as my original lab had no heating. I worked on other projects in the new room. When I finally had time to begin experiments with the universal indicator, I could not find the small bottle for quite a while. Over the course of six months, I had forgotten where many things were in my old laboratory. When I eventually found the bottle, I was overjoyed. 
If larger beakers had been used (for example, 1 or 3 liters), the color difference between the two samples would have been more pronounced, but this was not the purpose of the experiment. I planned to add a small amount of universal indicator powder to each beaker. At that moment, a colleague entered the room. Taking the opportunity, I showed him the beakers and asked whether he noticed any difference in color between the liquids. He did not know what they contained. After some thought, he remarked that the liquid in the second beaker was slightly yellowish, while the first was colorless. Thus, he independently confirmed the observation. Incidentally, this was the same colleague who had given me the universal indicator six months earlier. I explained my intended procedure. He objected, noting that the solid indicator should first be dissolved in alcohol before being added to water; otherwise, the experiment would fail. I thanked him and followed his advice. I placed a small amount of universal indicator powder into a glass and added a few milliliters of alcohol. A reddish-brown solution formed. I then added a few drops of this solution to each beaker and stirred. The distilled water turned yellow, while the tap water turned green. A faint yellow tint is visible in the video, although I did not notice it during direct observation. According to the comparison scale, the pH of the distilled water was approximately 6, while that of the tap water was around 8. Both beakers were then placed on an electric hotplate and heated to boiling for several minutes. The color of the distilled water did not change significantly, whereas the yellowish-green color of the tap water became clear green. According to the comparison scale, this corresponded to a pH of about 9. In other words, the pH increased by approximately one unit, although the color change was far less pronounced than in the phenolphthalein experiment. After several days, the color of the solutions in both beakers remained unchanged. __________________________________________________ 1 Phenolphthalein has several color transition ranges. In an alkaline medium, phenolphthalein is fuchsia; however, in a highly alkaline medium (pH > 12), it becomes colorless again. In slightly alkaline, neutral, and acidic environments, the indicator is colorless; however, in a highly acidic medium (pH < 0), phenolphthalein turns orange. However, for many practical purposes, it is the color transition of phenolphthalein at pH 8.2 and above that is significant, as is the case for the experiments described in this article. Фенолфталеин имеет несколько интервалов перехода. В щелочной среде фенолфталеин малиновый, однако, в сильнощелочной среде (pH > 12) он снова становится бесцветным. В слабощелочной, нейтральной и кислой среде индикатор бесцветный, зато в сильнокислой среде (pH < 0) фенолфталеин становится оранжевым. Однако, для многих практических целей имеет значение именно переход окраски фенолфталеина при pH=8.2 и выше, равно как и для описанных в статье экспериментов. 2 Hydrolysis (tap water) [link]. |
|
Вода и универсальный индикатор - Часть 2
Как известно, для чистой воды pH=7. Растворы кислот имеют pH < 7, а растворы щелочей имеют pH > 7. В первом случае концентрация ионов водорода выше, чем в чистой воде, а во втором - ниже. Если pH одного раствора на одну единицу меньше, чем pH второго раствора, то концентрация ионов водорода в первом растворе в десять раз выше, чем во втором.
Идеально чистая вода имеет нейтральный pH, который точно равен 7, однако природа не любит идеальных вещей. Например, водопроводная вода обычно слабощелочная из-за гидролиза растворенных в ней солей. Следовательно, pH водопроводной воды обычно немного выше 7. Дистиллированная вода практически не содержит растворенных солей, зато обычно содержит растворенный углекислый газ, который попадает в воду из воздуха. Углекислый газ образует с водой слабую угольную кислоту, поэтому дистиллированная вода имеет pH ниже 7. Например, при измерении pH дистиллированной воды были получены значения: 6.5, 6.3 и даже 5.5. Чтобы удалить из воды углекислый газ, ее долго кипятят, затем охлаждают в закрытой колбе, соединенной с атмосферой трубкой, заполненной гидроксидом бария. Индикатор фенолфталеин имеет pH перехода окраски примерно 8.2. При pH < 8.2 фенолфталеин бесцветный (кислые, нейтральные и слабощелочные растворы), а при pH=8.2 и выше индикатор приобретает малиновый цвет [1]. Если в один стакан налить водопроводную воду, а в другой дистиллированную воду и добавить в оба стакана несколько капель раствора фенолфталеина, индикатор останется бесцветным. Если теперь нагреть стаканы на электрической плитке, то дистиллированная вода все равно останется бесцветной. Зато водопроводная вода вскоре приобретет слабую малиновую окраску, которая будет усиливаться по мере нагревания. Кипящий раствор уже будет интенсивно-малиновым. Нагревание усиливает гидролиз солей, а также вызывает разложение гидрокарбонатов с образованием карбонатов, что приводит к возрастанию рН - раствор становится более щелочным. В результате достигается точка перехода фенолфталеина, и раствор становится малиновым. После охлаждения воды начинается обратный процесс, однако, происходит он далеко не моментально. В моем эксперименте [2] окраска фенолфталеина исчезала очень медленно. Даже через три дня был заметен слабый малиновый оттенок раствора. Возникла идея повторить данный эксперимент, используя вместо фенолфталеина универсальный pH индикатор. Лирическое отступление. Я занимаюсь химией примерно 38 лет. Еще с детства мне приходилось иметь дело с универсальной индикаторной бумагой - точно с такой, какая показана на фотографиях выше (см. первую часть статьи). Однако, универсальный индикатор в виде порошка в бутылочке я встречал всего несколько раз. Компоненты универсального индикатора pH сравнительно недорогие и доступные, но обычно его выпускают в форме индикаторной бумаги. В прошлом году коллега подарил мне одну такую бутылочку с универсальным индикатором. Я положил ее в ящик стола, чтобы не потерять, и запланировал провести с индикатором эксперимент. Однако, из-за зимних холодов мне пришлось перейти в другую лабораторию, поскольку моя тогдашняя лаборатория не имеет отопления. В новой комнате я занимался другими работами. Когда появилось время, чтобы начать эксперименты с универсальным индикатором, я долго не мог найти крохотную бутылочку. За полгода я успел забыть, где что лежит в старой лаборатории. Когда таки нашел бутылочку - сильно обрадовался. 
Если использовать стаканы большего объема, например, 1 или 3 л, то разница окраски двух образцов воды станет более заметной, но такой цели не было. В каждый из стаканов я собирался добавить немного порошка универсального индикатора. Неожиданно зашел коллега. Пользуясь случаем, я показал ему стаканы и спросил, заметил ли он разницу между цветом жидкостей? Коллега не знал, что в них находится. После раздумий коллега сказал, что жидкость во втором стакане чуть желтоватая, а в первом - бесцветная. Таким образом, он независимо подтвердил мое наблюдение. Кстати, это был тот самый коллега, который подарил мне универсальный индикатор полгода назад. Я объяснил ему, что собираюсь делать. Коллега возразил, что твердый индикатор нужно сначала растворить в спирте, а только потом добавлять в воду, в противном случае эксперимент не получится. Я поблагодарил коллегу и сделал, как он порекомендовал. Насыпал в стакан минимальное количество порошка универсального индикатора и добавил несколько миллилитров спирта. Образовался красно-коричневый раствор. Добавил несколько капель этого раствора в каждый из стаканов, перемешал воду. Дистиллированная вода окрасилась в желтый цвет. Водопроводная вода приобрела зеленый цвет, причем на видео заметен слабый желтый оттенок, а при непосредственном наблюдении я его не наблюдал. Согласно шкале сравнения, pH раствора дистиллированной воды составлял около 6, а водопроводной воды - около 8. Поставил оба стакана на электрическую плитку, включил нагрев. Кипятил растворы несколько минут. Цвет первого стакана (дистиллированная вода) существенно не изменился, во втором стакане (водопроводная вода) желтовато-зеленый раствор стал зеленым. Согласно шкале сравнения, цвет стакана с водопроводной водой теперь соответствовал рН около 9. Другими словами, рН воды увеличился примерно на 1, но изменение окраски выглядело далеко не так наглядно, как в эксперименте с фенолфталеином. После нескольких дней стояния цвет воды в стаканах не изменился. |

Water and Universal Indicator |

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

Glacial acetic acid |

|

|

|

Hailstones |

|

|
|
Комментарии
К1
У меня где-то были миниатюрные ампулы с универсальным индикатором, но там вроде уже спиртовой раствор.
К2 Когда я учился в школе, бывала такая "диверсия": на стройке в бочку с разведённой известью добавляли таблетку фенолфталеина аптечного, толчёную. К3 Мне встречалась еще как минимум один вариант выпуска универсального индикатора- готовый спиртовой раствор в бутылочке. В детсве мне достался набор Н.И. Алямовского, он предназначался для определения кислотности почвы. набор состоял из эталонов окраски индикатора в запаянных пробирках, визуального колориметра, баночки с индикатором и еще некоторых мелочей. Самое класное, что было в этом наборе, - карболитовый клевый ящик И еще в догонку фотография оригинальной упаковки индикатора из статьи.  |